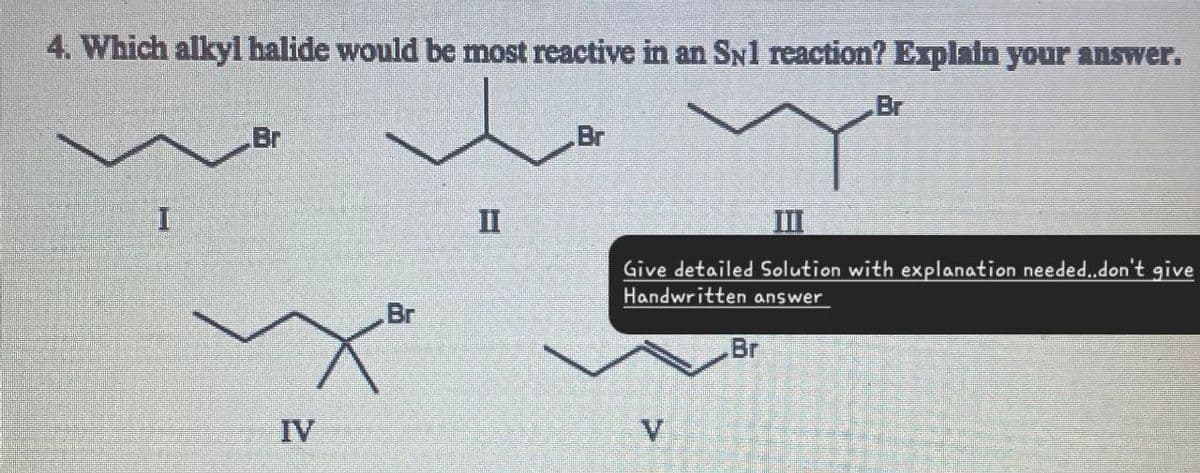
4. Which alkyl halide would be most reactive in an SN1 reaction? Explain your answer. I Br IV Br Br Br II III Give detailed Solution with explanation needed..don't give Handwritten answer Br V
Q: Draw the products of this reaction. Ignore inorganic byproducts. На сво HO- -H HO-H HOH HOH CH2OH a…
A: Kiliani-fischer synthesis is a method to increase the length of carbon chain of carbohydrates.It…
Q: For the following molecule, enter the number of carbon and hydrogen atoms in the spaces provided.…
A: In a bond line structure, carbon -carbon bonds are represented using lines.The intersection point of…
Q: The equilibrium constant, Kc, for the following reaction is 1.80×10-2 at 698 K. Calculate Kp for…
A: Equilibrium constant is defined as the ratio of concentration of products to the concentration of…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic acid side…
A: Given reaction:We have to draw the major product of this reaction.
Q: (please correct answer and correct and incorrect option explain) For autosomal dominant disease with…
A: The objective of the question is to understand how to differentiate between carriers of an autosomal…
Q: 35.5 g O, in a 15.7 L container at 1.25 atm ☐ K x10
A: Given that,Mass of Molar mass of Volume of the gas= Pressure of the gas in the container=…
Q: Problem 13 of 14 Submit A common ketone starting material is shown below. Predict the major product…
A: Answer shown in attachment provide in explanation box Explanation:
Q: Fill in the missing information in the following table. pH POH a. Solution a 9.67 b. PH рон Solution…
A:
Q: Use appendix E : Thermodynamic data from p. 203 workbook, to calculate the enthalpy of formation…
A: Given:Carbon monoxide (g) reacts with hydrogen gas to produce carbon tetrahydride (g) and water…
Q: Draw the pyranose Haworth projection from the Fischer projection shown below. H HO-H HO-H HOH HO H-…
A: Given Fischer projection of a monosaccharide. Draw the pyranose form of Howarth projection.
Q: 4. Determine the theoretical heat of dissolution of sodium hydroxide using the heats of formation…
A:
Q: kc-R' Ketone CH₂ CH₂- - CH₂ CH₂ 10: ון R-CH Aldehyde + CH3CH₂-- CH₂CH 3 - OH + CH3 CH2 - Ộ - CH2 CH3…
A: The question is based on the concept of solubility. solubility follows a basic principle "like…
Q: In the reaction below, the concentration of the nucleophile stays the same while the concentration…
A: SN1 reaction rate depends upon the concentration of alkyl halide only.Rate of SN1 is in this order…
Q: Incorrect Your answer is incorrect. For the reaction below: 1. Draw all reasonable elimination…
A:
Q: Question 15 Draw major E2 ELIMINATION product. Hint: Review Chapter 10.3, the elimination can only…
A: This reaction is an example of E2 reaction is which final product is always an alkene.
Q: a. Predict the 13C NMR (# of peaks, integration of each peak, and relative chemical shift of each…
A: The signals in the NMR based on the chemical environments of the atoms present in the compounds.If…
Q: Identify the expected major product of the following Diels-Alder reaction. <.
A: The objective of this question is to choose the correct major product obtained from the given diels…
Q: Problem 8 of 14 Submit Curved arrows are used to illustrate the flow of electrons. Using the…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Draw a structural formula for the product of the reaction shown. CH3 NC CO₂CH3 H H CH3
A: The objective of the question is to identify the product.
Q: Considering rotation around the bond highlighted in red, draw the Newman projection for the most…
A: The objective of the question is to draw the Newman projection for the most stable and least stable…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A: I do not know the precise flow chart on page 167 (Chapter 10.9) that you referred to in the…
Q: Calculate the solubility at 25 °C of CuBr in pure water and in a 0.0140M CoBr, solution. You'll find…
A: Given that,The concentration of solution =Ksp value of Molar mass of Then,
Q: The volume of a sample of hydrogen gas was decreased from 13.82 L to 7.11 L at constant temperature.…
A: Given,initial volume of hydrogen gas = 13.82 Lfinal volume of hydrogen gas = 7.11 LFinal pressure…
Q: For the reaction below: 1. Draw all reasonable elimination products to the right of the arrow. 2. In…
A: When an alkyl halide reacts with a strong base like NaOH then elimination reaction happens. The…
Q: Which of the following is the correct definition for an Arrhenius acid? O a. electron pair acceptor…
A: The objective of the question is to identify the correct definition of an Arrhenius acid from the…
Q: エー
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: write the balanced equation for an acid base reaction that would produce each of the following…
A: An acidic solution and a basic solution react together in a neutralization reaction that forms a…
Q: Draw a structural formula for the product of the reaction shown. CH3 CH3 • • Use the wedge/hash bond…
A: This is an example of Diels-Alder reaction. In this reaction a conjugated diene undergoes…
Q: Hypoiodous acid (HIO) is used as a strong oxidizing agent. The K₂ of HIO is 2.3x10-11 at 25°C. What…
A: Hypoiodous acid (HIO) = 0.874 MKa of HIO is 2.3×10-11 at 25°C. Calculate the pOH of hypoiodous acid.
Q: Which statements are true about weak acids? They dissociate completely in water. The Ka values are…
A: The objective of the question is to identify the correct statements about weak acids among the given…
Q: Question 4 Draw Zaitsev and Hofmann products that are expected when each of the following compounds…
A: E2 elimination is a type of elimination reaction in organic chemistry where a proton and a leaving…
Q: Draw the major organic product of this E1 elimination reaction. Ignore byproducts. 0.01 M NaOH heat…
A:
Q: For each of the following strong base solutions, determine [OH−],[H3O+],pH, and pOH. Determine…
A: The objective of the question is to determine the concentration of hydroxide ions [OH−], hydronium…
Q: Write the empirical formula for at least four ionic compounds that could be formed from the…
A: A ionic compound is formed by the electrostatic interaction between the cation and the anion. The…
Q: E. Calculating thermodynamic quantities (Individual Work) Temperature (°C) Temperature Ksp (K) AG°…
A: The equilibrium constant, temperature, enthalpy, and entropy are related according to the following…
Q: The penultimate (next-to-last) step of a Wolff–Kishner reduction involves an E2 step. Predict the…
A: The Wolff-Kishner reduction is a chemical reaction used to convert carbonyl compounds like ketones…
Q: Experiment 12 Report Sheet (continued) XeF $ C₂HzBr CS2 IF 5 C103 Molecule Dot Structure Bonding…
A: The objective of the question is to determine the dot structure, bonding sketch, electron pair…
Q: Draw the monomers that could be used to form the following polymers.( don't use hand raiting please…
A: Monomers are small molecules that join to form more complex molecules called polymer in a repeated…
Q: For each of the following strong base solutions, determine [OH−],[H3O+],pH, and pOH. Determine…
A: The objective of the question is to determine the concentration of hydroxide ions [OH−], hydronium…
Q: H₂C H₂C Write the products of your compound and the following reagents. If the reaction would not…
A: Aldehydes react with to get oxidized to carboxylic acids.
Q: Provide the product for the following reaction. OH 1. NH₂NH2/ H* 2. KOH/H₂O/A 3. H3O+ OH OH NNH2 OH…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Write the correct IUPAC name for the compound below CH, OH CH₂CHCH₂CHCH₂CH B) 5-methylhexan-2-ol A)…
A:
Q: Q write mechansim Two reactions: (1) 1., H (1 equiv.) 2. a) LiAlH b) H2O work - up 3. H (final…
A: The objective of the question is to write the mechanism for the given reactions. However, the…
Q: A buffer contains significant amounts of acetic acid and sodium acetate. Enter the complete…
A: The objective of the question is to understand how a buffer solution, which contains significant…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the phthalic acid…
A: The reaction given is,
Q: Draw the dicarbonyl starting material of the reaction shown below. Ignore inorganic byproducts.…
A: Sodium borohydride, NaBH4 is a reducing agent that can reduce a carbonyl functional group into an…
Q: The thermal conductivity of Cl2 at 298 K and 1 atm is 8.9×10-3 J. K-1.m¹.s-1 Part A What is the…
A: Thermal conductivity of Cl2 = Temperature = 298 KPressure = 1 atmEnergy gained by the molecule…
Q: Some soluble compounds are listed in the table below. Complete the table by filling in the name or…
A: Part I.Part II.Explanation:Part I.Ionic compounds consist of positive (metal) and negative ions…
Q: Self Study/Carboxylic Acids/Synthesis/Reactions Name: 1. Show how you will accomplish the following…
A:
Q: K Problem 12 of 14 Submit Draw the missing organic structures or select the missing reagents in the…
A: Starting Materials:PhCHO (phenyl ethanol)H3O+ (hydronium ion)PBr3 (phosphorus tribromide)DMF…
Step by step
Solved in 3 steps

- Does ether cleavage following SN1 or SN2 ? Give your explanation.Please answer the attached question. Give the major products for the appropriate SN1 and E1 reactions and please explain how you calculated your answers. Thanks.12c. If you could, please provide an explaination for why the major product would be the SN2 product rather than E2 product?
- The following compound readily eliminates CO; to form a conjugated six membered ring. (a) Complete the reaction with drawing the possible structure and (b) explain why this reaction goes steadily.what is the order of reactivity by ranking from most to least reactive in SN2 reactions?For SN1 Explain if 3o ( tertiary) alkyl halides reacted (fastest or slowest) explain why. Be sure to explain if alkyl halides did not react or did react and why. 3o (tertiary) compounds listed are: 2-chloro-2-methylpropane (see picture) Base your explanations on the following considerations: the nature of leaving group, the effect of structure, steric hindrance and any other feature.
- Please complete both subpart reactions in clear handwritten answer on a paper!How do you properly write an SN1 reaction, with transition states/intermediate steps included?Explain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.

