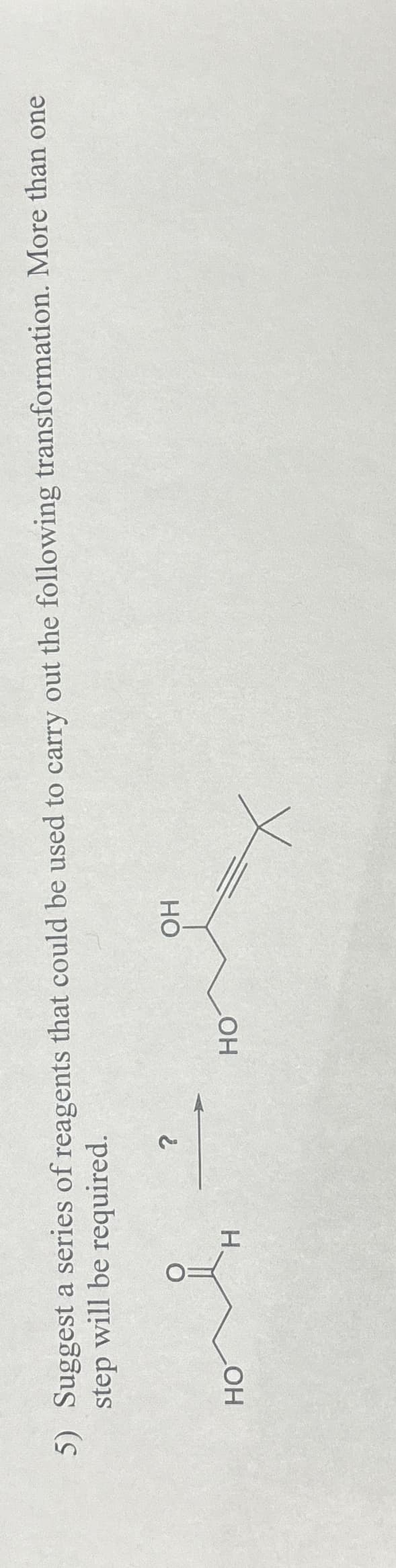
5) Suggest a series of reagents that could be used to carry out the following transformation. More than one step will be required. ? HO H OH HO
Q: Calculate the amount of heat (in kJ) required to convert 79.6 g of water to steam at 100 °C. (The…
A:
Q: :Which of the following are Enantiomers
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: A 1. CH>MgBr 2. H₂O B 0 D 1. KCN 2. H₂O 1. CHILI 2.H₂O* 1. (CH3)2CuLi 2. H₂O* ©
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: Predict the products of this organic reaction: + H2O + HCI ? N Specifically, in the drawing area…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: What percentage of aqueous phenylammonium ion is deprotonated at pH = 8.530? The Ka of phenyl…
A: The pH is given as 8,530Ka M
Q: Calculating the pH at equivalence of a titration 0/5 Izabella A chemist titrates 80.0 mL of a…
A: pH = 5.90Explanation:Given: V(CH3)2NH=80.0mL;[(CH3)2NH]=0.7536M;[HBr]=0.0940M;pKb=3.27at the…
Q: H₂N´ Edit Drawing CH3 Edit Drawing OH NH H2O, H₂SO4 (cat.) OCH3 (1) CH3CH2MgBr (excess) (2) H3O+
A: Cyclic amode also known as lactam .on hydrolysis it breaks to acid and amine group.
Q: Ultraviolet radiation has wavelengths from 1.0×10-8 to 1.0×10-7 m, whereas the wavelength region for…
A: The objective of the question is to compare the frequency and speed of ultraviolet radiation with…
Q: Dinitrogen trioxide decomposes to NO and NO2 in an endothermic process (DHo = + 40.5 kJ/mol)? N2O3…
A: 1) Right 2) Right 3) Left 4) Left 5) LeftExplanation:The given reaction is N2O3⇌NO2+NOand…
Q: 1 +
A: In the given organic species, the carbon adjacent to a carbonyl group contains a negative charge.…
Q: 12 10 Hd 8 6 0 0 4 8 12 16 20 24 28 32 36 Volume Base Added (mL) 8A. What was in the Erlenmeyer…
A: A buffer is a solution which resists any change in pH on adding a small amount of acid or base.It is…
Q: Which of the following statements best describes the role NaOH plays in the synthesis of aspirin?…
A: The question is asking about the role of sodium hydroxide (NaOH) in the synthesis of aspirin.…
Q: caq1 Organic Chemistry problem. Please help with. Thanks
A: The main product formed is given belowExplanation:
Q: PART 1: For the decalin derivative below please identify the correct Newman projection: Please type…
A: The planar decalin structure is converted to chair structure and then easily the Newmann projection…
Q: Suppose a 500. mL flask is filled with 0.40 mol of CH4, 0.30 mol of H₂S and 0.10 mol of CS2. This…
A: The objective of the question is to find the initial molarity, change in molarity, and equilibrium…
Q: Complete the mechanism for the heterolysis step.
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas…
A:
Q: The following reaction would yield: CH₂CH H&C H&C CH₂OH Oa. An optically active product Ob. No…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Fill in the left side of this equilibrium constant equation for the reaction of hydrofluoric acid…
A: [HF][H3O+][F−]=Ka Explanation:
Q: At-9.93 °C the concentration equilibrium constant K = 6.2 for a certain reaction. Here are some…
A: The objective of the question is to answer the questions related to the information provided by the…
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A:
Q: Draw Zaitsev and Hofmann products that are expected when each of the following compounds is treated…
A: The reactant is an organic compound. It is a secondary alkyl halide where the halogen is a bromine…
Q: draw all possible structures for electrons in 3d orbitals
A: for d-orbitals, l=2Hence, m=-2,-1,0,1,2on the basis of probability calculations there are 5 possible…
Q: H₂C H NBS CH3 CCl4, hv H3C Br CH3 **You may assume that Br-Br is formed by a side reaction that…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Provide the correct name for the compound below. Answer: Answer OH
A: Here, a structure of an organic moleucle is given, We have to give name of the molecule.
Q: Give the product of the reaction.
A: The alkyl nitrile gives the hydrolysis reaction in the presence of an acid or base catalyst to form…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting structure,…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: In 10.0 mL of caffeine in water sample, 20.0 mL of 0.1M iodine solution and 2.0 mL of sulfuric acid…
A: The reaction between Caffeine and Iodine is as follows:Given,
Q: Which group r compo Ma Mag This
A: magnesium reacts with oxygen and forms magnesium (II) oxide Charge on Mg = +2Charge on O = -2
Q: Draw the major organic product(s) of the following reaction. CI H₂O + NaOH You do not have to…
A:
Q: 20. Which of the following salts will be substantially more soluble in acidic solution than in…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas…
A: Given,width of a stainless-steel cylinder = 20.0 cmlength of a cylinder = 24.0 cmmass of a carbon…
Q: 2. If the compound of interest had to be distilled under reduced pressure, what modification could…
A: Distillation is a fundamental technique in chemistry used for separating and purifying substances…
Q: All the following reactions follow the law of conservation of mass except--- 4CO2 + 2H2O 2Fe2O3…
A: Find out the reaction those who do not follow the law of conservation of mass
Q: A sample of NaOH weighing 0.40 g is dissolved in water and the solution is made to 50.0 cm³ in…
A:
Q: Mass of CaCO3 standard (g) Volume of CaCO3 used (mL) Volume of EDTA Trial 0.15099 34.90 Trial 2…
A: The % mass of a molecule in a given mixture can be calculated using the following equation-
Q: Show all significant resonance contributors and a resonance hybrid for the following molecule.
A: When all the properties of a molecule cannot be explained by a single structure, we draw more than…
Q: Payalben
A: The objective of this question is to calculate the specific heat of a substance given the mass of…
Q: Br H₂C H H₂C Br Br. A D G CH₂ H,C. Br B CH₂ E Br CH₂ H Br. H₂C .. CH₂ F Br Br CH₂ To answer it…
A: To find out the most stable conformation of the given substituted cyclohexane first we shall draw…
Q: Below are two reactions, and one will occur faster than the other. Ts. O TS. NaCN DMSO CH3 Reaction…
A: The objective of the question is to predict which of the given reaction will occur faster.The polar…
Q: Draw the major organic product(s) of the following reaction. CI + NaOCH3 CH₂OH
A:
Q: Is it Endo or Exo?
A:
Q: A student obtained the following data from this experiment. Calculate weight of the gas vapor in…
A: Mass of flask and foil cap = 89.617 gMass of flask, foil cap, and condensed vapor = 91.458 gFlask…
Q: a.) List all of the possible values for l in the 1st energy level (n = 1). Leave fields blank if…
A: The question has two parts. The first part is asking for the possible values of the angular momentum…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A: Alkyl halides undergo E2 elimination reactions in the presence of a strong base. The elimination…
Q: Give the major product of the following reaction. Br₂ hy O There is no reaction under these…
A: The required answer is given below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: HF C14H20
A: Given reaction refers to the replacement of an aromatic proton with an alkyl group. This is done…
Q: A reactant (N2O5) in a first order reaction decreases to a conc. of .011 M over 40s. The rate…
A: The objective of the question is to find the original concentration of the reactant N2O5 in a first…
Q: Question 4. Predict the most likely site of electrophilic aromatic substitution in each compound…
A: The electron density at the aromatic ring determines the rate at which the electrophilic aromatic…
Q: Carbon disulfide gas and oxygen gas react to form sulfur dioxide gas and carbon dioxide gas. What…
A: Carbon disulfide gas (CS2) reacts with Oxygen gas (O2) to produce Sulfur dioxide gas (SO2) and…
Step by step
Solved in 3 steps with 1 images


