C7H12O4 I need help answering 2 3 4 5 For IHD please show step by step and I need to know ir analysis: absorption band if it there is a ring or pi bond,frequency and label peaks on the graph
C7H12O4 I need help answering 2 3 4 5 For IHD please show step by step and I need to know ir analysis: absorption band if it there is a ring or pi bond,frequency and label peaks on the graph
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
ChapterL4: Proton (1h) Nmr Spectroscopy
Section: Chapter Questions
Problem 23CTQ
Related questions
Question
C7H12O4
I need help answering 2 3 4 5
For IHD please show step by step and I need to know
ir analysis: absorption band if it there is a ring or pi bond,frequency and label peaks on the graph

Transcribed Image Text:HIT-NO-1424 SCORE-
DIETHYL MALONATE
SDBS-NO-1419
IR-NIDA-04418 : LIQUID FILM
LO0
50
D
4000
3000
2000
1000
IAVENUMI ERI -l
3639 84
3561 81
3465 79
2986 23
2943 44
1763
11 16 36
32
1416 34
788 72
684 74
1742
1393 35
1371
1333
1097
1735
13
1037
676 70
1478
48
10
966
6B
692 68
586
1467
39
1271
13
941
6B
68
2910
53
1457
49
1191
13
857 64
846 62
2878 68
1448
30
1162
10
#29 (300 MHz)
triplet
не
singlet
4H
quarkat
10
8
7
6
4
3
2
HPH-00-299
ppm

Transcribed Image Text:DU (IHD): Show calculation:
2.
Comments:
Report IR analysis here. Tabulate
3.
Report your 1H NMR analyses here: Tabulate, chemical shift, splitting, neighboring protons,
integration*, fragments, etc.
Group chem.Shift,leen Integration Splittng tof Neigle His Fragments
Conclusion:
Draw Structure and match the peaks with the protons.
5.
4.
Expert Solution
Step 1
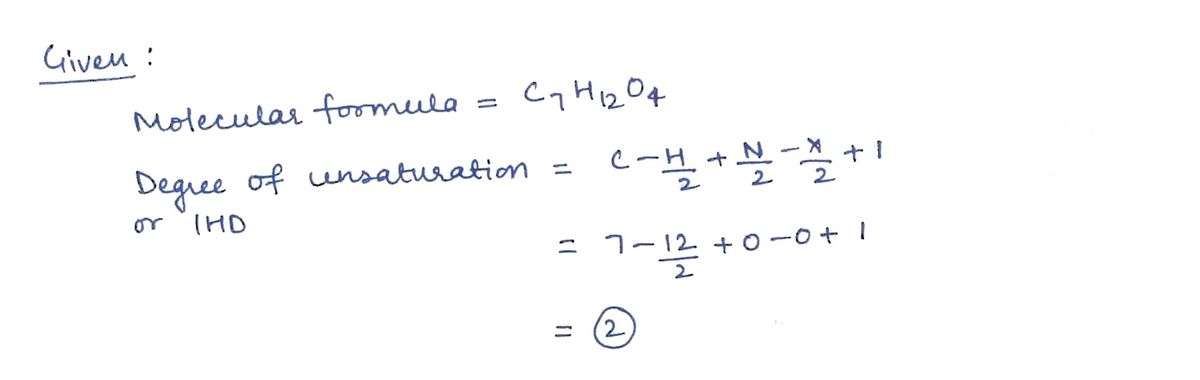
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole