World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter13: Gases
Section: Chapter Questions
Problem 49A
Related questions
Question
Evaluate the Fraction (Please see screenshot)

Transcribed Image Text:Evaluate the fraction.
(6.38 × 104) (5.15 × 106)
2.51 x 108
1.31
Incorrect
< Feedback
Macmillan Learning
Although you can simply enter the
numbers in your calculator and let the
calculator do the work, some
calculators can be confusing in the way
that exponential numbers are entered. It
is always good to do a mental check in
your head, estimating the order of
magnitude of the answer, to make sure
you have not made a mistake in
entering the numbers.
In evaluating the fraction, treat the
coefficients and the powers of 10
separately at first. Remember to add
exponents when multiplying and to
subtract exponents when dividing.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
Hi, thank you for the explanation. Although I understand what you are referencing, I placed that response and it said incorect.
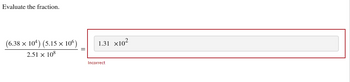
Transcribed Image Text:Evaluate the fraction.
(6.38 × 104) (5.15 × 10°)
2.51 x 108
1.31 ×10²
Incorrect
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning