Fe (aq) (a) Fe(s) + Cu" (aq) (b) Mg(s) + Cl2(8) (c) 2 Al(s) + Cr,0;(s) → 2 Cr(s) + Al,O;(s) + Cu(s) MgCl2(s)
Fe (aq) (a) Fe(s) + Cu" (aq) (b) Mg(s) + Cl2(8) (c) 2 Al(s) + Cr,0;(s) → 2 Cr(s) + Al,O;(s) + Cu(s) MgCl2(s)
Chapter1: Numerals And Fractions
Section: Chapter Questions
Problem 5RP
Related questions
Question
Identify the oxidized reactant, the reduced reactant, the oxidizing agent, and the reducing agent in the following reactions:

Transcribed Image Text:Fe (aq)
(a) Fe(s) + Cu" (aq)
(b) Mg(s) + Cl2(8)
(c) 2 Al(s) + Cr,0;(s) → 2 Cr(s) + Al,O;(s)
+ Cu(s)
MgCl2(s)
Expert Solution
Step 1
The electron accepting molecule or ion is known as oxidizing agents. As the electrons are accepted it causes the oxidation of another species. The electron donating species is known as reducing reagents. By donating electron, it causes the reduction of other species.
Step 2
Increase in oxidation number – oxidation
A decrease in oxidation number – reduction
(a)
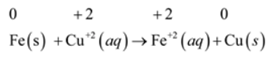
- The oxidation number of Fe increased 0 to +2 i.e. Fe is oxidized. Thus Fe is the oxidized reactant.
- The oxidation number of Cu decreased from +2 to 0 i.e. Cu+2is reduced. Thus Cu+2 is a reduced reactant.
- The reduction of Cu+2 to Cu is caused by the Fe. So Fe is the reducing agent.
- The oxidation of Fe to Fe+2 is caused by the Cu+2. Thus, Cu+2 is the oxidizing agent.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

