MasteringChemistry: CHE154 S X This Mac can't connect to iCloud b a problem with "ayanaboo10@gma A session.masteringche Open iCloud preferences to fix this probler Later iCloud Prefe
Q: What mass of NaCH3COO should be added to 1.00 L of 0.100 M CH3COOH to produce a solution with pH =…
A: Buffer is a solution that resists the change in pH when small amount of acid or base is added to it.…
Q: Calculate the pH of a solution formed by mixing 15.0 mL of 0.250 M HCIO with 15.0 mL of 0.315 M…
A: Answer :- The pH of solution formed by mixing 15.0 ml 0.25 M HClO with 15.0 ml 0.315 M NaClO = 7.64…
Q: A 250.0ml of a solution of the strong base, CsOH(MW = 150.0 g/mole) is made. The pH is measured to…
A: ***As per Bartleby's guidelines, I can answer only the first main question. You can resubmit the…
Q: The pH of a 0.24 M solution of dimethylamine is 12.51. Calculate the Kb value for dimethylamine (4.4…
A: Hello. Since more than one question has been asked, the first question will only be solved in this…
Q: MasteringChemistry: CHE154 X +…
A: Given information: Concentration of C6H5CO2H = 0.461 M Ka for C6H5CO2H = 6.5 x 10-5
Q: Oxalic acid is a diprotic acid that occurs naturally in some plants. Calculate the pH and the…
A:
Q: = Report - Goog X ed Scholarship Ap CovalentActivity.do?locator-assignment-take NGLSync Porta OWLv2…
A:
Q: What is the resulting pH of a solution at 298 K made by mixing 50.0 mL of 0.355 M HCl and 25.0 mL of…
A:
Q: My Grades - BIOL111ASP2020 MB Canvas Login at Mary Baldwin Ur X O Chemistry 121 101 Chem101 Question…
A: A strong acid shows complete dissociation to respective anion and H3O+whereas a weak acid can only…
Q: Calculate the pH of a solution prepared by mixing 0.0870 mol of chloroacetic acid plus 0.0250 mol of…
A: pH =pKa + log[Conjugate Base][acid] pH =pKa + log[Sodium Chloroacetate][Chloro acetic acid] pH…
Q: MISSED THIS? Read Section 18.2 (Pages 788 - 799) ; Watch KCV 18.2B, IWES 18.2, 18.3. A 100.0-mL…
A: Given: A 100.0 mL buffer solution contains 0.175 M HClO and 0.150 M NaClO.
Q: A solution is made by dissolving 43.5 g of Ba(NO₂)2 in 500.0 mL of water. (a).As NO₂ is a base,…
A:
Q: What volume of 0.0500 M sodium hydroxide should be added to 250 mL of 0.100 HCOOH to obtain a…
A: Weak acid react with base form salt ,if acid is in axcess then it form buffer solution
Q: Calculate the pH of the solution that results when 32.0 mL of 0.1980 M NH3 is (This problem requires…
A:
Q: Hello, i am provided the pH levels and volumes for Bromocresol Green weak acid by strong base and…
A: The table can be set up as follows in order to plot a graph:
Q: A S by disson 19 27.5 g HONH3CIO3 in 500.0 mL of water. Using Ka(HONH3*) = 9.1 × 10-7, determin the…
A: Calculate molarity first and then use √Ka×C PH=-log[H+] Molarity is defined as total number of…
Q: Calculate the pH of a solution prepared by mixing 0.0870 mol of chloroacetic acid plus 0.0250 mol of…
A:
Q: For the following questions, wherever is necessary write the chemical equation(s) (pay attention to…
A: a) HCOOH dissociates as: HCOOH⇌HCOO-+H+ Construction of ICE table: HCOOH⇌HCOO-+…
Q: For the buffer solution of weak acid HA and its conjugate base salt NaA, [HA] and [A] are expressed…
A: ka and kb are the dissociation constant .As this value is very small,in the range of…
Q: The pH of 280 mL of a 0.188 M solution of unknown weak base is 11.76. What is the Kb of this…
A: Determine the pOH of the weak base.
Q: Calculate the percent dissociation of crotonic acid (C, H,CO,H) in a 0.53 mM aqueous solution of the…
A: The term percent ionization can be defined the fraction of the weak acid or base which ionizes in…
Q: Calculate the pH of 100.00mL of 0.20 M HCIO4 solution after 124.00 mL of KOH 0.20 M have been added.…
A: Given that: Volume of HClO4 = 100.0mL = 100.0ml×1L/1000ml = 0.1L Molarity of HClO4 = 0.20M Molarity…
Q: What is the pH of a solution that is prepared by dissolving 6.78 grams of acetic acid (formula…
A: Given information: 6.78 grams of acetic acid The molar mass of acetic acid is 60.05 grams/mol 8.74…
Q: Under a 25°C subsurface environment, the groundwater has reached equilibrium with soil minerals and…
A: Given: pH of ground water = 7.4 pKsol = 8.3; Ksol = 10- pKsol = 10-8.3 = 5.01 x 10-9 CaCO3…
Q: MasteringChemistry: CHE154 S X session.masteringchemistry.co This Mac can't connect to iCloud I a…
A: Concentration of OH- formed during the dissolution of NaF in water is determined as follows,
Q: 11 5, de /188795/assignments/3601808 Homework Ch16 - Attempt 1 Problem 16.61 3 of 15 Constants |…
A: Given- Concentration = 0.13M pKa = 2.32
Q: Name: Date: Section: Prelaboratory Assignment: pH: Measurement and Application. 1. Calculate [H']…
A: We are given three different pH and we have to find the concentration of H+ and OH- ion. PH =…
Q: (from the in class activity) - Suppose you dissolve three tablets of Sudafed (R), each containing…
A: It is a salt made from weak base and strong acid. So, pH = 0.5pKw - 0.5pKb - 0.5logC ...(1) Total…
Q: What is the pH of a solution that contains a concentration of 1.35 x 10−5
A: Given concentration = 1.35 x 10-5 M
Q: IP wc dissolve an a cid weak HzA in wate, the solution volume willbe looml, and its pH=27 if you…
A: The question is based on the concept of chemical equilibrium. We have to calculate mass of the acid…
Q: nemist dissolves 405. mg of pure sodium hydroxide in enough water to make up 80. mL of solution.…
A: We will use formula of pOH .
Q: Calculate the volume (ml)of concentrated ammonia and the weight (gm) of ammonium chloride…
A: Calculating the pOH of the buffer by using the Henderson-Hasselbalch equation: pOH=pKb + log…
Q: For Practice 17.11 - Enhanced - with Feedback Part A Find [OH- for a 0.026 M solution of LIOH.…
A: Metallic hydroxides (MOH) tend to dissociate wholly into their ions and therefore, regarded as…
Q: O.235g of weak benzoic acid CHC6Hfc02) is dissolve el in of solation. A solutionwith o.l08 M NGOH IS…
A: The pH of a solution is used to determine the acidity or alkalinity of a solution. The pH is given…
Q: Calculate [OH ] and pH for each of the following solutions. (a) 0.0096 M LIOH [OH] = 0.0096 M pH =…
A: pH and pOH are related with each other pH + pOH =14 pOH = - log[OH-] Higher the value of -OH ion,…
Q: Calculate the pH of each of the following buffered solutions. Ka of acetic acid = 1.8 x…
A: Since you are asked for multiple sub parts, we are entitled to solve first three only.
Q: Formic acid owes its name to the fact that some ant (fourmi) species have specialised soldiers that…
A: First we will draw IRE table ,then using it we will calculate pH of solution.
Q: Part A What is the pH of a buffer solution prepared by mixing 20.0 mL of 0.0000 mol L NaOH with 20.0…
A: Volume of NaOH solution used in buffer solution = 20 mL Volume of caodylic acid used to prepare…
Q: What is the expected pH of a 150.0 mL 0.250 M solution of HOCI which is diluted to a 500.0 ml mark…
A: Given that, a 150.0 mL of 0.250 M solution of HOCl is diluted to 500.0 mL, i.e., V1 = 150.0 mL, S1 =…
Q: 1. At 50°C, find the pH of (a) pure H20 ( Kw = 5.47 x 101“), (b) 0.100 M HCI, (c ) 0.200 M NAOH, (d)…
A:
Q: A chemist titrates 230.0 ml. of a 0.6154M ammonia (NH,) solution with 0.6407M HNO, solution at 25…
A:
Q: 36. Lactic acid is a weak acid with the formula HC3H5O3, the Ka for lactic acid is 1.38 x 10-4 In…
A: Given, HC3H5O3 + H2O ⇔ C3H5O3- + H3O+ The Molarity of Lactic acid = 0.2931 M and it's volume = 250…
Q: HE154 S20 Ch17 Sec7-9 Later iCloud Preferences... r Practice 17.12- Enhanced - with Feedback • Part…
A:
Q: Calculate the volume (ml)of concentrated ammonia and the weight (gm) of ammonium chloride…
A:
Q: I am unsure on how to solve this problem, and it requires not use the H+ approximation where it…
A: Hydrolysis of HCl Being a strong electrolyte complete dissociation of HCl takes place.…
Q: Dashboard 10 Chem101 b Answered: At a particular tempe + i app.101edu.co Question 8 of 23 Submit How…
A:
Q: An alkaline solution was prepared with LiOH in such a way that 0.65 g dry LiOH was directly mixed…
A:
Q: For H2S, Ka1 = 9.6 10-8, and since Ka2 = 1.3 10-14, 0.04 M Na2S Calculate the pH of the solution by…
A: Given, Ka1 = 9.6×10-8 and Ka2 = 1.3×10-14

Methylammonium chloride dissociates into quaternary ammonium ion (CH3NH3+) and chloride ions (Cl-). Chloride ion (Cl-) is the conjugate base of a strong acid so it will not influence the pH of the solution. Methylammonium ion is the conjugate acid of a weak base, so an acid-dissociation reaction determines the pH of the solution.
The balance equation is given below,
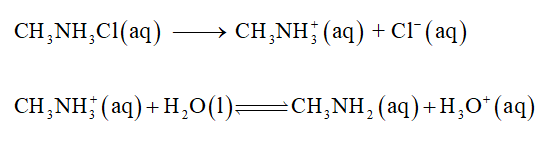
Ka value of CH3NH3+ is calculated,
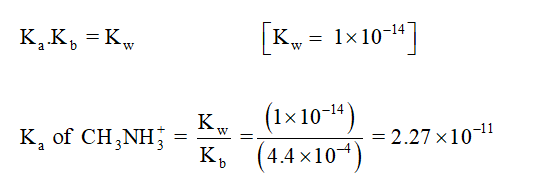
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- AutoSave 20200930175438D1747_Assigment 5 Equilibrium Compatibility Mode - Word P Search Gleyren Zefanya O X ff ?ם) File Home Insert Design Layout References Mailings Review View Help Acrobat A Share P Comments Find - Calibri (Body) v11 - A A Aa v A AaBЬСcDd AaBbСcDd AaBbC AаВЬСс. АаВ LG Paste E Replace BIU v ab x, x A vev A I Normal 1 No Spac. Heading 1 Heading 2 Title Create and Share Request Dictate Sensitivity Editor A Select v Adobe PDF Signatures Clipboard N Font Paragraph Styles Editing Adobe Acrobat Voice Sensitivity Editor 3. 5.. 6.|7. 8. 1 10. 1 11. 1 12. 1 13. 1 14. 15. I 16. 1 17. | 18. 1 1. Write equilibrium expressions for each of the following equilibria: (a) 2 C(s) + O;(g)22 CO(g) (b) Zn²*(aq) + H,S(g)2ZnS(s) + 2 H*(aq) (c) HCl(g) + H,O(e)2H;O*(aq) + Cl^(aq) (d) H;(g) + O,(g)2H,O(g) 2. The equilibrium constant for the reaction, 3 H;(g) + N2(g) 22 NH;(g), at a given temperature is 1.4 x 10-7. Calcu- late the equilibrium concentration of ammonia, if [H2] = 1.2 × 10-2 molL-…nd engy Level above Ferm; Level if prebabl.tg f enrgy levelis o03 and temportun is 6ookMe 03, -78 °C then Zn, CH;CO2H
- 3 eq req For the reaction N₂ (9)+ 3H2(g) → 2NH3 (9) AH = -92.2 kJ and AS = -199 J/K The equilibrium constant for this reaction at 256.0 K is Assume that AH and AS are independent of temperature. Submit Answer $ R References to access important values if needed for this question. Retry Entire Group 9 more group attempts remaining Show Hint Cengage Learning | Cengage Technical Support 7 Z U * 8 1 M ( K O L Previous Next> Save an 5Ca Fz at cquálioriun c Naf and is is 1. a) wyht chemical equation ( balanced) cenilibrim r reachoon explession (K for reacion ar durease and whiy increase मपत् (० d) will Ca or durcase innd whyOWLV2 | Online teaching and lea X 9 New Tab + x ow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator3Dassignment-take m Week 1 Discussion... MasteringAandP: Q.. P MasteringAandP: S... [References] Use the References to access important values if needed for this question. In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used to determine the specific heat of a solid, or to measure the energy of a solution phase reaction. A student heats 60.94 grams of silver to 98.31 °C and then drops it into a cup containing 79.27 grams of water at 21.78 °C. She measures the final temperature to be 25.06 °C. The heat capacity of the calorimeter (sometimes referred to as the calorimeter constant) was determined in a separate experiment to be 1.69 J/°C. Water- Assuming that no heat is lost to the surroundings calculate the specific heat of silver. Metal- sample Specific Heat (Ag) J/g°C. %3D Submit Answer Try Another Version 2 item attempts remaining EM…
- What is the major organic product of the following reaction? H,0/ heat HO2C. .co.CH .co.H HO,C HO HOC (A) (В) (C) (D)Chrome File Edit View History Bookmarks People Tab Window Help A ALEKS - Jacqueline Hoppenrey x C Search Textbook Solutions | Ch x G standard formation reaction for x G convert kj to j - Google Search x + A www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IQİHQRDYV_6Ux63SypJXz0Coxvwqgg4JkWI7IUnyRjVNsBjUR5oMNI9R1NdBbh5VEqEfXiSb O THERMOCHEMISTRY Writing a standard formation reaction Write a balanced chemical equation for the standard formation reaction of solid vanadium(V) oxide (V,0s). Explanation Check 2021 McGraw-Hill Educ Reserved TermsM req For the reaction E 20 H₂(g) + Cl₂(g) → 2HCl(g) AH = -185 kJ and AS = 20.0 J/K The equilibrium constant for this reaction at 261.0 K is Assume that ΔΗ° and AS are independent of temperature. Submit Answer SA 4 000 000 R F Show Hint DE LO % 5 Retry Entire Group [Review Topics] [References] Use the References to access important values if needed for this question. T G Cengage Learning Cengage Technical Support < C 6 9 more group attempts remaining Y H & 7 U 8 J I 9 K 0 0 P Previous Next Email Instructor Save a +
- Non-Commercial Substances Hypothesis: Soluble in Test Bond Molecular Structure ΔΕΝ H2O? Result: H20 Solubility Result: Hexane Substance Classification Solubility Hexane? H2O: pS Sodium Na--Br lonic Bromide 1.9 Hexane: I |--| lodine NP C 0.0 Ammonium N/A lonic Carbonate NH4* O O NH4* Sodium N/A lonic Acetate C--H NP C 0.4 H-C-OH C--O PC H-C-OH Glycerol 1.0 H-C-OH O--H PC 1.4 HIA ALEKS - Jacqueline Hoppenrey x C Search Textbook Solutions | Ch x G standard formation reaction for x A www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IQiHqRdYV_6Ux63SypJXz0Coxvwqgg4JkWIZIUnyRjVNsBjUR5oMNI9R1NdBbh5VEqEfXiSbDcv772zARqYw50M O THERMOCHEMISTRY Calculating a molar heat of reaction from formation enthalpies Jacc Using the table of standard formation enthalpies that you'll find under the ALEKS Data tab, calculate the reaction enthalpy of this reaction under standard conditions: Fe,O3(s)+3 CO(g)→2 Fe(s)+3 CO,(g) Round your answer to the nearest kJ. Explanation Check 2021 McGraw.Hill Education All Rights Reserved Temms of Uss Privacy Accessic MacBook Air IICalculate the equilibrium constant of the reaction given the following thermodynamic data: H+ (aq) + ОН- (аq) — Н20 () H+ (aq) OH- (aq) Н20 () AfH°, kJ/mol S°, J/(K-mol) -230 -285.8 -10.9 70

