MasteringChemistry: CHE154 X Smart Pearson Player Smart Pearson Pla session.masteringchemistry.com/myct/itemView?assignmentProblemID=D1 CHE154-H Gen
Q: eterm 18) Major groduct for rxn: NaOH H20 20) Correct reggents peeded? EtO OEt a) ) NaOMe 2) ETOH b)…
A:
Q: Saved Normal BIIIU X2| X² | → EIEIE 田=| IT| 图|三| |li If you had a mixture of butyric acid and…
A: The question is based on the concept of experimental chemistry. these are the methods which are used…
Q: It CHCH3 z)z R or S 1 8omer
A: The conformation given is,
Q: x. The oxidation shown below. 9 ..0 PCC = Ci-Cr-o: H-N But for the mechanism you can use chromic…
A: Introduction: The oxidation of alcohol to aldehyde can takes place via pyridinium chlorochromate…
Q: In cach reaction box, place the best reagent and conditions from the list provided. 1) Br 2) 3) 4)…
A:
Q: What reagents can be used to this picture? Choices : Potassium permanganate, Pyridinium…
A:
Q: What is the predominant form of EDTA at pH 5? a. H3Y- b.H2Y2- c.HY3- d. Y4-
A: Full form of EDTA hexa ethylene diammine Tetra acetic acid. PH= 5 means it is Acidic solution . As…
Q: Match the reaction to their product. Fe(NO3)3 + NH4OH White precipitate Fe(NO3)3 + KSCN Red colour…
A:
Q: 2. What is the species responsible for the positive result in Fehling's Test? Complete the table…
A:
Q: 4. HCHO + KMNO4 +_(4a)_ → _(4b)_+ K2SO4 +| MnO24 + H2O 5. CH3CH2ONA + CH3CH2B1 15) + NaBr. 6.…
A: Balance and complete the reactions.
Q: What is the key equation for spectrometry experiments? O q_rxn = -q_cal O d = m/V O A = ebC O at…
A: Answer:- this question is answered by using the simple concept of spectrometry in which the…
Q: DIBAL, -78°C toluene 5. 2. Hot
A: Diisobutylaluminium hydride is a reducing agent with the formula (i-Bu₂AlH)₂, where i-Bu represents…
Q: Compound AG°F, kJ/mol S°, J/mol-K PCI3 -286 312 CH3CH2OH -175 161 CH3CH2CI -53 190 H3PO3 -232 82
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: MasteringChemistry: CHE154 x Sat Poarson Player Smart Pearson Player MasteringChemistry: CHE154 P…
A: For the given reaction : Initial concentration of COCl2 = 1.6 M Equilibrium…
Q: 1. catramolecular D-A 2. then refro-D-A (loss co,) 4. co, Me intramolecular かA 5. Intramoka culne…
A:
Q: +2-1 014 salentt ligand prepared by condensation of salicylaldchyde (I equiv.) and ethylenediamine…
A:
Q: LOD NH 500 L000 4 D00 3000 2000 1500 HAVENUMB ERI 1l 50 4 D00 3000 2000 15 00 1000 5 DO HAVENUMB ERI…
A: IR spectroscopy helps us to find the functional group in the given compounds .
Q: Predict the solubility and explain why: a) CdCO3 b) Na2S c) PbSO4 d) (NH4)3PO4 e) Hg2Cl2
A: The maximum amount of solute that can be dissolved in the definite amount of solvent at a fixed…
Q: Which of the following reactions would have a positive AS" ? (i) Na₂SO4(s)--> 2 Na*(aq) + SO4² (aq)…
A: Entropy order -> S(gas) > S(liquid) > S(solid) ->Hence , if number of gaseous moles…
Q: Which equation has a AHxn that is not equal to APf of the product? Multiple Choice 02(g) + H2(9) →…
A: The heat of reaction (∆Hrxn) is given as the difference in heat of formation of reactants and…
Q: What mass of chionium cil) chloride cISp:3S almol) is pralwed by thereaction of 39.69 chbine with 15…
A: When a chemical reaction takes place with more than one reactant then the concept of limiting…
Q: (b) [Zn(II)(H2O)6]?* can undergo successive reactions with phen ligands: [Zn(II)(H2O)«(phen)]²* +…
A: Phen ligand (b) as number of phen ligand increases stability (logKf) decreases due to increase…
Q: File Edit View History Bookmarks Profiles Tab Window Help * O D $ D Fri Sep 17 ••• I Course Home…
A: When Given acetic acid is treated with methoxide ion , then esterification Reaction take place and…
Q: ОН 1. TBSCI а. imidazole, CH2Cl2 A DMF, -18 °C to rt 2. NalO4, CH2Cl2 80% НО OH large excess
A: We have to give the product of the given reaction. We know that the TBSCl is used to protect the…
Q: Exercice 3: Equilibrer les équations suivantes Ni H2SO4 H2 Niz(SO.)3 a. +] -> b. NH4F AlCl3 NHẠCI…
A: The complete balanced equation : b. 3NH4F + AlCl3 →3NH4Cl +AlF3 c. 3Na2CO3 + 2H3PO4 →2Na3PO4 +3H2O +…
Q: 1. _________ FeBr3 + ______H2SO4 -----> _____Fe2(SO4)3 + ____HBr Show Work
A: All things are provided in handwritten solution.
Q: Find New equation 2me 3 m 00 Peyo, t co lel-2e )co, ia Smd Cn 2 m Fe Look for the conversion factor…
A:
Q: base* = K₂CO3 or CaCO3 L = ligand biaryl product (~ 92%)
A:
Q: EXP 20 Report Sheet Heats of Formation Name Wet Lab: a.Is the reaction in the flask warm or cold? Is…
A: Soln:- Given reaction NH4Cl(s)---------> NH4+(aq) + Cl-(aq) Since NH4Cl is dissociating into ions…
Q: K2Cr,0, H2SO4 Compound Tollen's Test Alkaline Iz in NaHCO3 Mg ribbon KI Colorless solution even…
A: Qualitative analysis is a stream of chemistry that comprises using both organic as well as inorganic…
Q: Vindow Help ) 51% Sun 10:46 AM session.masteringchemistry.com g... Class Schedule Listing Schedule…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: as SN1, SN2, El or E2. Mal
A:
Q: Make a reasonable prediction stating whether the molecule will be soluble in water or not and…
A:
Q: O Resources LX Give Up? V Hint Check Answer on 9 of 10 Reaction B. Draw the products of Reaction B.…
A:
Q: What are the products of the following reactions?
A: Since you posted question with multiple sub-parts, we are entitled to answer the first 3 only.
Q: / to loml of 0.01M A9NO3 was added lond afo-o2 M Naz Soy - Show by calculet whether or not…
A: To know whether the product will precipitate or not, the value of reaction quotient (Q) should be…
Q: Predict the product .CH3 KMNO4 H20 Br HO HO. Br Activ Option 1 Go to Option 3
A:
Q: ny nickel atoms a MM (P) = 30 97
A:
Q: + AIATS For Two Year Medic... 90 /180 (02:53 hr miny Mark for Review HC = CH red hot Fe tube 873K…
A: When acetylene gas is passed through red hot iron tube, it results in the formation of benzene…
Q: (1) The value of AS° for the catalytic hydrogenation of acetylene to ethene, C2H2 (g) + H2 (g) →…
A: Given thermodynamics Reaction is - C2H2(g) + H2(g) ----> C2H4(g) For this Reaction,. Entropy…
Q: home Comment Edit Page Protect 102.33% 14/14 ol 1 D D Q O Rotate 目 D- Edit Text Edit Picture PDF to…
A: Interpretation: Starting from either benzene or phenol how MON-0585 can be synthesized is to be…
Q: of Remove water NH catalytic H30* pyrrolidine
A: In this question we have to tell the product of the reaction.
Q: Excess CH3OH H3O* PCC Br TART HERE 1) Mg 2) CH3CHO 1) 2) 6) ? Br KMNO4 DIBAL , 7) 8) H3C SOCI2…
A: The given compound is bromo benzene. Here we have to find out the products when various reagents are…
Q: Give a clear handwritten answer and Explain...?
A: The given statement is true. As It can be oxidized by losing two protons and two electron to form…
Q: diluted H2SO4 "ОН ОН checkpoint-08v02-toc-ay21-22fa.pdf (page 18 of 18) OH OH 'OH "OH racemic…
A: The first reaction occurs via acid hydrolysis reaction in which addition of H-OH takes place while…
Q: Fill out the chart below Compound Weight in Mol. Wt. Moles Melting pt. in 1,4-diphenyl-1,3-butadiene…
A: A question based on mole concept that is to be accomplished.
Q: Compound Addition of FeCl3 Observations Dark ben on top botton Separation of both colors Phenol…
A: ASPIRIN: Aspirin is also known as acetylsalicylic acid. The molecular formula of aspirin is C9H8O4.…
Q: 1. Ignoring Steveochemistry draw the streetere for all passion different nonobromnation prodocts Brz…
A: Hello. Since more than one question has been posted, the first question shall only be answered in…
![MasteringChemistry: CHE154 X
Smart Pearson Player
Smart Pearson Pla
session.masteringchemistry.com/myct/itemView?assignmentProblemID=D1
CHE154-H Gen
<CHE154 S20 CH16 Sec6-9
Chapter 16 Multiple Choice Question 24
Part A
Calculate the value of [N2leg if [Hleg = 1.5 M, [NH3]eq
0.5 M, and K. = 2.
%3D
=
%3D
N2(g) + 3 H2(g) =2 NH3(g)
0.15 M
0.17 M
0.062 M
0.037 M
0.019 M
Submit
Request Answer
Provide Feedback](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F8ddcfb62-a75f-441b-9ba0-61b722369038%2F1d4ad4cb-c634-4b5d-bb9f-6d1f7609a113%2Fx9j5yjb.jpeg&w=3840&q=75)
First, the Kc equation for the given reaction is determined.
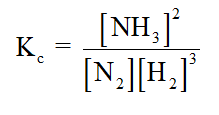
Step by step
Solved in 2 steps with 2 images

- a) CI Ph3PlI. - PPH3 Rh PPH3 + H2 ? Ph3P CO b) Mn2(CO)10 + HCl c) CH3I CO CO CH4 d) Co2(CO)s + H2 CO e) [Fe(CO),(CH;)]* + CH;CH,Br [기 → ? f) H A +PPH3 PPh3 Ph3P g) EtgPl.pt- + LICH3 + PHCH,Br EtgPNernst equation for pair IO3 |I': RT E = E° + nF In a(10;)a°(H*) a(I¯) RT a(IO;)a(H") E = E° + nF a(I") RT E = E° –- nF In a(IO; )a°(H*) a(I¯) nF a(IO,)a°(H*) lg RT E = E° + a(I¯)ALEKS AlecNema - Learn O Liquid octane CH3CH26CH3 will x A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-lgNslkr7j8P3jH-lvUrTNdLZh5A8CnG03PBGuXr8iCPa7ZMmymzAt1HXJMLs6azoqH3wiGm1Zuh4j2Za9PeCgDhi. Apps Sprouts Academy:. 9 Online Tutoring C 400 Request Heade.. O Weather & Soil C.. O STOICHIOMETRY Limiting reactants CH,(CH, and gaseous water (H,O). Suppose 3.4 g of Liquid octane (),CH;) will react with gaseous oxygen (o,) to produce gaseous carbon dioxide (CO,) octane is mixed with 23.0 g of oxygen. Calculate the minimum mass of octane that could be left over by the chemical reaction. Round your answer to 2 significant digits. Check O 2020 McGraw-Hill Education, All Rights Reserved. Terms of Use Privacy Acces Explanation O Type here to search
- Nernst equation for pair Cr,0,|2Cr": RT a(Cr,0; )a4(H*) a°(Cr³*) E = E° + In nF RT a(Cr,0} )a(H*) lg nF E= E° + a²(Cr³*) RT E= E° + nF lo9(Cr,O; )a'* (H*) a(Cr³*) nF E = E° + RT lo a(Cr,O; )a'*(H*) a(Cr³*)is polar or nonpolar. Do not draw double bonds to oxygen atoms unless they are needed for the central atom to obey the octet rule. Do not include formal charges in your drawing. XeO3 polar nonpolar 826RRA 000-F 12TScholarsh X = Report - X Grammar X ent/takeCovalentActivity.do?locator=assignment-take 2req = 2req =2req 5 SM M S s2req O ts 2req ts M (M) ots ots O NGLSync X OWLv2 | X Cengage X * 0 Use the References to access important values if needed for this question. What is the calculated value of the cell potential at 298 K for an electrochemical cell with the following reaction, when the H₂ pressure is 7.74x10-3 atm, the H+ concentration is 1.42M, and the Ni2+ concentration is 4.99 × 10-¹M? 2H+ (aq) + Ni(s) → H₂(g) + Ni²+ (aq) Ecell = V The cell reaction as written above is spontaneous for the concentrations given: Submit Answer Retry Entire Group 9 more group attempts remaining a #3 O C 73°F E 56 0 Update 1:43 PM 5/27/2022 Ę X
- /x/Isl.exe/lo_u-IgNslkr7j8P3jH-IQs_dp5pR4ENzvdYC-70kXyMz36BqJhw3sVPj_jpaFLxvGArYxlbmayqa71YYPJBG6RjdYAdGPjGhFLILID-HEX1YcqAB?1oBw7QYjlbavbSPXtx-YCjsh_7mMmrq#item P Course Home Login | Student Veri... Hb Logout MyProgrammingLab Imported From IE O ITEC2110:Summer2... TunesToTube - Upl.. Publix App Web Development.. O KINETICS AND EQUILIBRIUM Using the Arrhenius equation to calculate k at one temperature fr. Cia 23%#3%# The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an activation energyE=45.0 kJ/mol. If the rate constant of this -1 -1 reaction is 4.4 x 10 M s at 266.0 °C, what will the rate constant be at 177.0 °C? Round your answer to 2 significant digits. -1 k = \\M -1 x10MasteringChemistry: CHE154 x Sat Poarson Player Smart Pearson Player MasteringChemistry: CHE154 P Pearson Sign In A session.masteringchemistry.com/myct/itemView?assignmentProblemID=142156078&offset%3Dprev CHE154-H Gen Chem II Bronikowski S20 Ayana Provide Feedback MacBook Air 44 II9:25 PM Fri Oct 7 x Syllabus 1 bonding Po a x syllabus 2 Isomeris... Br Untitled (Draft) ✓ X orgo practice exam 1 T X 3.5+ 3.6 76% Untitled (Draft) what is the proper name of thefe maecules? using traynham namendatue -pls also include stereochemistry Pir nescending. nescessary. |

