Q: A sticky substance that upon dissolving in water, is found to be a nonelectrolyte, has a concentrati...
A: Molarity=Number of moles of solute/volume of soln in Lmolality=no. of moles of solute/mass of solven...
Q: If energy is required to form monatomic ions from metals and nonmetals, why do ionic compounds exist...
A: Ionic compounds are exist as crystal lattice and as not simple ion pairs.
Q: A 0.16-g sample of a purified protein is dissolved in water to give 2.0 mL of solution. The osmotic ...
A: Osmotic pressure for dilute solution, π = CST π = nVST where, π = Osmotic pressure C = concentrati...
Q: (a) Determine the oxidation number of lead in each of the following oxides: PbO, PbO2, Pb2O3, Pb3O4....
A: Oxidation number is defined as the number that is assigned to an atom when it loses or gains electro...
Q: How many kilojoules are released when 8.2 g of water condenses at 100°C and cools to 15 degrees Cels...
A: Given , 8.2 g of water condenses at 100°C and cools to 15 degrees Celsius m= 8.2 g Ti= 100°C Tf=15°C...
Q: The balanced equation for the decomposition of water is shown below. 2 H2O → 2 H2 + O2 If 0.72 g of ...
A: SO ACCORDING TO THIS BALANCED EQUATION 36 GRAMS OF WATER WILL DECOMPOSE INTO 4 GRAMS OF HYDROGEN GAS...
Q: Which of the following represents the correct balanced chemical equation for the burning of coal whi...
A: When coal is burned, it reacts with the oxygen in the air. This chemical reaction converts the store...
Q: how many moles are in 46.5 g of pure aluminum and how many atoms are in 46.5 g of pure aluminum?
A: 1. Mole in terms of mass: 1 mole = atomic mass of an atom. No. of moles (n) of an atom can be calcu...
Q: 204 + Dye → 2 CO2 + Excited Dye wo glow sticks are placed in water baths, one in hot water and the o...
A: Collision theory is used to predict the rates of chemical reaction particularly for gases. It is bas...
Q: Please answer everything pooooo this can really help me :(
A: "Since you've asked multiple questions, we'll solve the first three parts for you and for the rest t...
Q: Ammonia gas can be prepared by the following reaction: CaO(s) +2 NH, Cl(s) → 2 NH3 (g) +H20(g) + CaC...
A:
Q: What is the line notation for each compound? ethanol methanol acetone pentane 1-propanol 1-butanol
A: Line notations represent structures as a linear string of characters. The Simplified Molecular-Input...
Q: Iron(III) oxide and hydrogen react to form iron and water, like this: Fe,O3(s)+3 H,(g)–2Fe(s)+3H,O(g...
A: A numerical problem based on equilibrium concept, which is to be accomplished.
Q: A patient needs 1.5 g of a solid must be given to the patient pe O 60 tablets
A: The dose of a drug: it is the quantitative amount taken by patient for the intended medicinal effect...
Q: Calculate the freezing point depression of naphthalene/biphenyl Find the molality of Naphthalene/...
A:
Q: 1. State the functional groups present. What suffix will be used in the IUPAC name for this compounc...
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: Write the chemical composition of the compound formed when ferric chloride is added containing both ...
A: The chemical formula of Ferric chloride is FeCl3 . Ferric chloride contains halogen i.e., chloride i...
Q: 8. Predict the major product for each reaction of triglycerides. These are not new reactions! You le...
A: Hydrogenation is the process of adding hydrogen atoms to unsaturated fats until it becomes saturated...
Q: An automobile engine provides 576 Joules of work to push the pistons and generates 2340 Joules of he...
A:
Q: To improve conductivity in the electroplating of automo-bile bumpers, a thin coating of copper separ...
A: a) The mass of copper has to be determined, The half-reaction can be written as, Cu2+(aq)+2e-→Cu(s) ...
Q: What is the number of formula units in a 9.72 mol sample of NH4NO3?
A: 1 mole of the sample contains 6.02 x 10^23 formula units
Q: I need help on 3 f-h
A:
Q: A solution contains 3.90 g of a nonvolatile pure hydrocarbon in 95 g acetone. The boiling points of ...
A:
Q: Question attached
A: Retrosynthesis: The retrosynthesis is a backwards synthesis of target molecule from simpler precurso...
Q: N2 (g) + O2 (g) =2 NO (g) Describe a step-by-step method to calculate the equilibrium concentration ...
A:
Q: Chemistry Question
A: In water, the sodium carbonate dissociates into two distinct stages. ...
Q: The following reactants will undergo a Robinson annulation reaction in the presence of base (KOH). D...
A:
Q: QUESTION 7 The following reaction is exothermic: CF2Cl2() - CF2Cg) Cg) Select the energy diagram tha...
A: The heat is released during the exothermic process, therefore the energy of the reactants in this pr...
Q: Which resonance form contributes more to the resonance hybrid? Explain briefly. N-H H- CH3 H-ö CH3 W...
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for ...
Q: Where are the nodes in the wavefunction for a particle confined to a box with 0 < x < a and n=...
A: Wave function for a particle in a box with 0 < x < a and n=3 No of nodes = to be determined. ...
Q: Calculate [OH−] for the following solution; [H3O+] = 8.87×10−9 M Is the solution basic at 25°C?
A: pH is negative logarithm of hydronium ion. Given, [H3O+] = 8.87×10−9 M
Q: I need full accurate and detailed answers.
A: Arrhenius equation is an expression that relates to the activation energy, temperature, and the rate...
Q: Write the steps of ‘Modern Adsorption Theory of Heterogenous Catalysis.’
A: The Modern Adsorption Theory can be well explained the overall basic steps involved in the heterogen...
Q: (S)-Adenosylmethionine (SAM) is a cosubstrate that is involved in biological methyl group transfers....
A: (a) The appropriate curved arrows for the given reaction are shown below:
Q: D) Explain your answer. This is the most important part. Please type on an attached page. A complete...
A: Boiling point direct proportional to intermolecular force.
Q: I need help with answering the second part of this problem.
A: Conformational isomers are those isomers which are formed by the free rotation around Carbon Carbon ...
Q: If the percent yield for the following reaction is 65.0%, how many grams of KClO3 are needed to prod...
A: The question is based on the concept of reaction Stoichiometry. we have been given a reaction and pe...
Q: What volume of ethylene glycol (C2H6O2), a nonelectolyte, must be added to 14.0 L water to produce a...
A: ∆Tf = Kf x Molality Molality = Number of moles of solute/Mass of solvent in Kg
Q: QUESTION 10 Measurements taken during the following reaction showed a concentration of carbon m...
A:
Q: Carbon-hydrogen bonds exhibit a range of different chemical reactivity that depends on molecular str...
A: The carbon atoms of organic compounds are classified by the number of other carbons directly bonded ...
Q: Sodium and chlorine react to form sodium chloride:2 Na(s) + Cl2( g)¡2 NaCl(s)What is the theoretical...
A: The limiting reagent in a chemical reaction is a reactant that is totally consumed when the chemical...
Q: You start the following reaction with 0.411 M N2 and 0.398 M H2. At equilibrium, you determine the c...
A:
Q: Write the structural formula and name of the organic product for the reaction between an alkyne and ...
A:
Q: Question attached
A: Given: The mass of glucose is 80 g. The mass of the solvent is 2Kg. We know that the molecular weigh...
Q: Complete and balance the following nuclear equations by supplying the missing particle: 1 1H + 11 5B...
A: The total mass number and proton: H11+B511,mass number=11+1=12The number of proton=5+1=6
Q: A sample of 3.929 g of polystyrene was dissolved in toluene and diluted to a total volume of 100.8 m...
A:
Q: How many peaks are present in the NMR signal of attached labeled proton?
A: Number of peaks correspond to the number of different type of protons present in the molecule.
Q: The protein ribonuclease A in its native, or most stable, form is folded into a compact globular sha...
A:
Q: A sheet of gold weighing 10.0 g and at a temperature of 18.0°C is placed flat on a sheet of iron wei...
A:
Q: Q13.58) How much dry solute would you take to prepare each of the following solutions from the dry s...
A: there are many concentration measurement terms such as molarity, molality and normality etc.
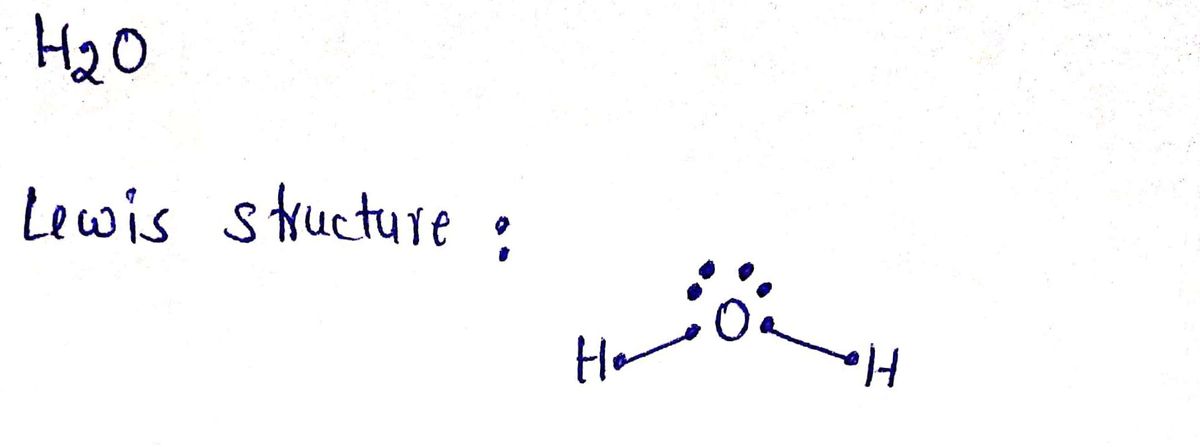
- Draw the lewis structures for H₂O, NH₃, CH₄
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- 10.) The structural formula of a certain aldehyde (related to formaldehyde) is H3C-CH2-CHO. Draw a Lewis structure for this aldehyde and determine the number of bonds present. Note that a single or a double or a triple bond counts as one bond. Write the number, not the word.Help me write the Lewis symbol for (Rn) pleaseWrite the Lewis symbols that represent the ions in ionic compound. K2O



