Q: How many grams of each ingredient is required to make 1500 g of the formula? Boric acid (5parts) Sta...
A: Given, Total mass of the formula = 1500 g Parts of each ingredients: Boric acid = 5 parts Starch = 1...
Q: Each of the chemical formulas is incorrect. Identify the mistake and rewrite the formula with the or...
A:
Q: Using your own words explain why ceramics are stronger and stiffer than polymers
A:
Q: just before a snow storm angelica poured salt on her drive way. why is salt applied onto driveway an...
A: We put salt on road before snowfall because it decreases the freezing point of water as a result the...
Q: NO is an atmospheric pollutant that destroys ozone in the stratosphere. Here is the accepted mechani...
A: Given reaction mechanism: We have to find the catalyst and the intermediate.
Q: A metallurgist needs to make 60 kg of architectural bronze, which consist of 12 parts copper, 1 part...
A:
Q: 1) Give the IUPAC names for the following compounds: (a) (b)
A: IUPAC nomenclature is used for naming organic compounds. Full form of IUPAC is international union o...
Q: Major Classes of Molecule Functional Example Ionic or How will the Group Polar? functional group aff...
A: Soln
Q: A solution of H2SO4with a molal concentration of 8.010 m has a density of 1.354 g/mL. What is the mo...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: The reaction of H2 with I2 is H2 + 12→ 2HI. Use reactant molecules to write appropriate elementary r...
A: Elementary reaction is the reaction which takes place in a single step. In elementary reaction, mole...
Q: When 60.0 mL of a 0.400 M solution of HN03 (ag) is combined with 60.0 mL of a 0.400 M solution of Na...
A: The volume of the final solution is = 60.0 mL + 60.0 mL = 120.0 mL The density of the solution is = ...
Q: Complete the following nuclear reactions and identify the cosmic phase (Big Bang Nucleosynthesis, St...
A: Answer - Nuclear reaction - Nuclear reaction is a process in which two nuclei, or a nucleus and an e...
Q: Consider the following reactions: A B, K,=6.63 A C, K2=2.00 What is K for the reaction C B?
A:
Q: Kinetics data were gathered for an enzyme-catalyzed reaction under identical conditions both with an...
A:
Q: Using the data in the table, calculate the rate constant of this reaction. A+B⟶C+DA+B⟶C+D Tri...
A: Order of reactions is sum of power of exponents to which molar concentration terms are raised in the...
Q: Make a suggestion of the reagents required to produce the following:
A: The functional groups of a molecule are a key component that plays a significant role in providing t...
Q: Consider the initial‑rate data at a certain temperature in the table for the reaction described by ...
A:
Q: crystalline or amorphous solid
A: A crystalline solid is formed by arranging the components in a regular repeating three-dimensional a...
Q: percent abundance
A:
Q: at mass of oxyg
A:
Q: 7. Compound X is an antibacterial drug of the sulfonamide group, and was widely used in the mid-20th...
A: Here we have to find the intermediate products formed during reaction sequence to prepare antibacter...
Q: OH H", heat HO H,0 H,0 Reduction [이 [이
A:
Q: 1. For the reaction below, determine the equilibrium concentration of all species. Kc=1x1020, [A]ini...
A: Given, Kc = 1x1020 [A]initial = 0.4 [B]initial = 0.6
Q: 2. The IR spectrum of an unknown sample shows a strong, sharp peak at 1725 cm1. Disregarding any oth...
A:
Q: how does an acidic cation interact with water? provide a net ionic equation for an acidic cation
A: Acid are those substances which gives H+ ions dissolved in water or in aqueous state.
Q: // IN B R tion 8 of 25 2 NaCl(aq) + K, S(aq) → Na, S(aq) + 2 KCI(aq) (3)°0E + (s)1D – (s)*(*01) O ac...
A: Introduction : Redox Reactions can be defined as the chemical reactions in which transfer of electr...
Q: Twenty-five mL of 0.100 M lactic acid (K, = 1.4 x 104) is titrated with 0.097 M KOH. a) What is the ...
A:
Q: b(NO2)2 is an ionic compound. What are the ions of which it is composed?
A:
Q: The reaction shown below is first order with respect to Fe when excess SCN" is present. The rate con...
A:
Q: _chloride ion bromide ion Lit F- Na* S2- K* Mg2+ Ca2+ 02- N3- P3- A3+ barium ion boron ion
A: While naming anion, 'ite' 'ate' or 'ide' suffix is used with the name of the element. While naming ...
Q: How much energy, in J, is required to raise the temperature of 1 kg of water from 37oC to 39.5oC? Th...
A: Given, The energy, in J, is required to raise the temperature of 1 kg of water from 37oC to 39.5oC i...
Q: If the initial concentration of N,05 is 0.60 M, what is the concentration of N2O5] after 2.0 hours? ...
A: Given, Initial Concentration of N2O5, [A]0 = 0.60 M Rate constant, k = 6.2x10-4 s-1 Time, t = 2 h...
Q: Hydrogen and oxygen react to form water, together they are a _____. - compound - molecule - mix...
A: When two or more types of molecules of different compounds are together then they will be a mixture....
Q: Sodium metal requires a photon with a minimum energy of 4.41 x10-19 J to emit electrons. a. What is ...
A: Given that - Minimum energy of emitted electron from sodium metal = 4.41×10-19 J. Then, Frequenc...
Q: . The acetate buffer contains 0.200 M acetic acid and 0.0500 M sodium acetate. Given that the Ka of ...
A: PH of buffer solution is calculated by using Henderson Hasselbalch equation
Q: 26. Calculate AG for the following reaction at 150 °C (attach your solution) C2H4(g) + 3 O2(g) 2 CO2...
A:
Q: NaSH Br 1) Hg(OAc)2, CH3CH2OH 2) NABH4 CH;CH,CH,Br
A:
Q: Write equations for the reaction of the buffer CH3COOH/CH3COO- buffer reacting with
A: In the buffer, Acidic part is reacts with base while Basic part reacts with acid.
Q: How to prepare the following compound by Retrosynthesis (RSA) and functional group interconversion (...
A: Synthesis of any target molecule can be explained with the help of retrosynthetic analysis of that p...
Q: A. Identify the name of the functional group where the given organic compounds belong (alcohol, ethe...
A:
Q: For the following reaction , the rate constant at 373 K i s 0.69856 min-1 . 1. Find the order of th...
A: Introduction: The order of the reaction can be determined from the unit of the rate constant. We hav...
Q: How many Liters are in 13.8 moles of CO2 gas @ STP? Group of answer choices A.13.8 moles x (1 L/22.4...
A: Here we have to find the volume occupied by 13.8 moles of carbon dioxide (CO2) at STP.
Q: a. Calculate the quantity of moles of NH3 gas that form when 4.20 mol of N2H4 liquid completely reac...
A:
Q: Determine each statement if it describes dissolution or disintegration. It is mainly affected by t...
A: Dissolution means the disove of substances in different solvents. It is applicable for solids, liqui...
Q: From your results, which component had the highest Rf value and which had the lowest? Based on your ...
A: The answer is as follows....
Q: Enter your answer in the box provided. What is the molality of a solution consisting of 56.1 mL of b...
A: Molality (m) :- The number of moles of solute dissolved in 1 kg of solvent is defined as molality ...
Q: A gas-phase reaction has the following four step mechanism: Cl2→2 CI CI+ CH4→ HCI + CH3 CH3 + Cl2→ C...
A:
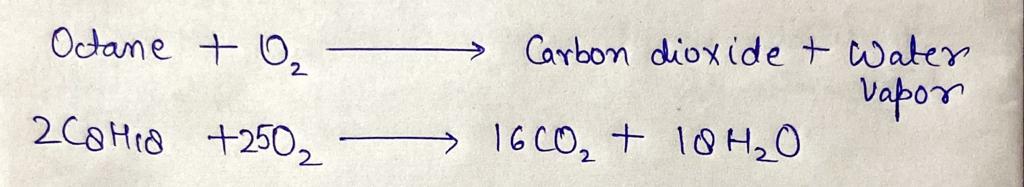
Q: uestions On complete combustion, one mole of an alkane produces two moles of carbon dioxide and thre...
A: Given: Moles of CO2 = 2 mol Moles of water = 3 mol
Q: An ionic bond is formed by the combination of positive and negative ions. (b) An ionic bond between...
A: Note: As per our guidelines, we are supposed to answer only the first question (i.e. part (a) to (i)...
Q: Using the IR table determine the functional group, identifying all signals that confirm the mentione...
A: IR tells us about the functional group present in compound .

Step by step
Solved in 2 steps with 2 images

- A ALEKS - Alec Nema - Learn G Complete the table below, which x C Complete The Table Below, Whici x + > C O A www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-lgNslkr7j8P3jH-IvUrTNdLZh5A8CnG03PBGuXr8iCPa7ZMmym9dT1il1Gkroy7AtUHLxV9FND2YCHj_luf5tjUsCtVPrkHH_G.. Apps Sprouts Academy. O Online Tutoring C 400 Request Heade. Q Weather & Soil CH. O THERMOCHEMISTRY Solving combustion thermochemistry problems Alec v On a 10 day wilderness expedition you'll need to heat 3.0 kg of water to the boiling point each day. The air temperature will average 25 °C. You have available canisters of compressed propane (C,Hg) fuel, which you'll burn to heat the water. Each canister has 50. g of propane in it. What is the minimum number of fuel canisters you must bring? The standard heat of formation of propane at 25 °C is -103.85 kJ/mol. You'll probably find other helpful data in the ALEKS Data resource. 圖 dlo canisters: Exliyanation Check O 2020 McGraw-Hill Education. All Rights Reserved. Terms of Use| Privacy |…T LI (271) Mann Bharryaa 2.0 - O ✔ X Netflix Type here to search = www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-lvdw7xgLCkqMfg8yaFKbD9GafJstkYLIJnusC-UibBjKoF3RIGVrvlxMuFtsCs1YV_jKNKNc6T1g4ASYj_65F27j0RiBMno?1o... Q O ELECTROCHEMISTRY Using the Nernst equation to calculate nonstandard cell voltage Explanation X TT ALEKS - Rafia Riaz - Learn A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 3+ 3 Cu²+ (aq) + 2Al(s) → 3Cu(s) + 2Al³+ (aq) 2+ Suppose the cell is prepared with 5.62 M Cu²+ in one half-cell and 7.99 M A1³+ in the other. Calculate the cell voltage under these conditions. Round your answer to 3 significant digits. 0 발 Check x10 + ロ・ロ Olo 0/3 Rafia V ? 圖 □ □ 图 olo Ar © 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility (?) 46°F Mostly clear C i 1:28 AM 5/5/2023 x M :K Haylee Scott - cp Zoom meeting lin com/web/viewer.html?state%=D%7B'ids %3A%5B"1zOflgo1GoQFrdviJ6PWx8L3h2j4uav5n"%5D%2C"action"%3A'open C Clever | Portal A Classroom https://sso.theleam. M Oro Grande Elemen. W Yearbook Aven ue Period 5 Scien. Haylee Scott - cprr061.pdf Building Vocabulary From the list below, choose the term that best completes each sentence. matter physical change endothermic reaction chemical change chemistry precipitate exothermic reaction 7. Any change that alters a substance without changing it into another substance is a(n) 8. is anything that has mass and takes up space. 9. A reaction that releases energy in the form of heat is called a(n) 10. A(n) absorbed. 11. A chemical reaction is also referred to as a(n) is a reaction in which energy is 12. A(n) a chemical reaction. is a solid formed from a solution during 13. is the study of the properties of matter and how matter changes. OPearson Education, Inc., publishing as Pearson Prentice Hall. All rights reserved…
- 00 %2A # 3. 5o09 awww.awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-lijulplKKWv4bwenmyrLYoBHBAAsJtmzj53GErnx_viTdHblGMF9jvgkSu_nrizREmdQXHPJbMzqOmOrXJcuonZITOhB66q?1oBw7QYlbav. LEKS-Courtney Wh New Tab + x O CHEMICAL REACTIONS Percent yield of chemical reactions Courtney S0 OC Gaseous butane CH. reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (CO,) and gaseous water (H,0). If 2.32 g of water is produced from the reaction of 1.74 g of butane and 10.2 g of oxygen gas, calculate the percent yield of water. Be sure your answer has the correct number of significant digits in it. % 固 Explanation Check ©2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibility 24 %23 2 4. 9. 5. 8. by k 4 69 C. alt 123ICE (SOs] = | M CH20] = 2 M • Construct (CE Table Given: %3D Find ko. SO3 6) + H,0 c9) ラH2SO4 ca)MoH t 7 Natt Cos, 7 three Cuive Hhe fell balance nequatiiys Hor He absove realtion?
- क Rb ALEKS Chemistry- 2021FA-CHI X A ALEKS - Griffin Barden - Learn eks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-JcZzdcvSCzsqTCIDqNGV3bKqhMfPmUcQ4EDeRİLhu6AbUmc3VVdLPcASZ3o67Wr4xcXIZmcLfpPf_PLSp4nj4wgn0SQA3P0?1oBw7QYjlI O THERMOCHEMISTRY Understanding the definitions of heat and work A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gaseous products. The 1 atm pressure cylinder has a piston that moves in or out, as necessary, to keep a constant pressure on the mixture of 1 atm. The cylinder is also submerged in a large insulated water bath. (See sketch at right.) The temperature of the water bath is monitored, and it is determined from this data that 259. kJ of heat flows into the system during the reaction. The position of the piston is also monitored, and it is determined from this data that the piston does 322. kJ of work on the system during the reaction. gases exothermic Is the reaction exothermic or endothermic? endothermic dn O…Ask Laftan An O st. John's Uni x A ALEKS - Iffat k x (SA Documents | F X M Inbox (132) (SA Aid Summary x SA Account Dash x O YouTube i www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-lijkPWvZoZLqKt1FLlq7wcPWKzBYGfE9IMFjNv26NxcUIFt8F81Y80MQxnZhc7Hg9N2QzofvWyND. ☆ O MATTER Iffat Calculating volume by combining the volume of simple shapes A chemistry student in lab needs to fill a temperature-control tank with water. The tank measures 28.0 cm long by 18.0 cm wide by 13.0 cm deep. In addition, as shown in the sketch below, the student needs to allow 2.0 cm between the top of the tank and the top of the water, and a round-bottom flask with a diameter of 10.5 cm will be just barely submerged in the water. Calculate the volume of water in liters which the student needs. Round your answer to the nearest 0.01 L. 2 cm flask water Explanation Check O 2021 McGraw-Hill Education. All Riahts Reserved. Terms of Use | Privacy I Acces 6. étv S MacBookk AirDetermine the hybridization of the central atom for each molecule. :ci: -AI- :CI: sp sp² sp³ O sp³d O sp³ d² -CI: sp sp² O sp³ sp³ d O sp³ d²
- 40 40 0.10 * 3:45 o an Anters eteat tar, Sar. tsiniac an Module 1 Snoe you a now tamilar with the parts and compositions ot the Periodie Tatia ot Elemerts, nowEs time tor us to know the chamical bonding ina nutaheil. We can now proceed to our nast actvity, dick this link impaw. younbe comvwatch?tko7RNSTE "Naming lonic Compounds" at wil hela you to anewer te activity. Analyze the figure below atd wite yout obeervation intide the box. 1OMISTS Figum 1 Figure 2 WHAT DO YOU SAY 1. Which figure represents Covalent Bondng? Why? z Which igum mpresents lonic Bandng7 Why? an Arterieal ania, In, en nrom Module 1 Nm Covn in sbamato. poarity a aaparation of ukecme IIo/index.html?deploymentld%3D55750828934189288909969212&elSBN=9781305657571&snapshotld%3D2199... Tp * NDTAP Q Search this co Use the References to access important values if needed for this question. Cyclopentasilane (SigH10) is a liquid with a density of 0.963 g cm3. It reacts with oxygen to give silicon dioxide (SiO2) and water. Calculate the mass of water that would form if 29.4 cm³ of cyclopentasilane reacted completely with excess oxygen. g water Submit Answer 4 question attempts remainingdit View History Bookmarks Profiles Tab Window Help ALEKS X Explanation ALEKS - Tiffany Te-L-Peng - L X + C www-awu.aleks.com/alekscgi/x/Isl.exe/10_u-IgNslkr7j8P3jH-liGBdp5ulp5VqUnMDyPOVaX6q0CRPLZSQ5NDHtEP4AK_R5q2H71rLJx4uFZRV O ORGANIC OXIDATION AND REDUCTION Predicting the products of alkene epoxidation Check Predict the major products of this organic reaction. Be sure you use wedge and dash bonds when necessary, for example to distinguish between major products with different stereochemistry. C C + T X RCO3H A S O $ 32 è Click and drag to start drawing a structure.

