Q/ Write complete pictorial electron configurations (lines and arrows) for each ion below. Do not use noble gas abbreviated configurations. Label the orbitals. Individual degenerate orbitals (e.g., px) do not need labels, but label the degenerate grouping. a) Mn2+ b) Ag+
Q/ Write complete pictorial electron configurations (lines and arrows) for each ion below. Do not use noble gas abbreviated configurations. Label the orbitals. Individual degenerate orbitals (e.g., px) do not need labels, but label the degenerate grouping. a) Mn2+ b) Ag+
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter2: Atomic Structure And Periodicity
Section: Chapter Questions
Problem 158CWP
Related questions
Question
Q/ Write complete pictorial electron configurations (lines and arrows) for each ion below. Do not use noble gas abbreviated configurations. Label the orbitals. Individual degenerate orbitals (e.g., px) do not need labels, but label the degenerate grouping.
a) Mn2+
b) Ag+
Expert Solution
Step 1
Mn+2 = 23 electrons
1s2 2s2 2p6 3s2 3p6 3d5 4s0
complete pictorial electron configuration :-
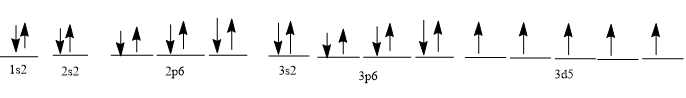
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning