
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 8.2, Problem 2P
Classify each
a.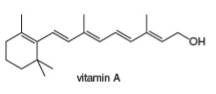 b.
b.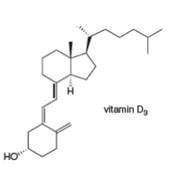
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Cyclic esters and amides are called lactones and lactams, respectively. These compounds are classified based on the carbon that is attached to the heteroatom of the ring. Classify the following compounds as a, b, g or d lactones or lactams
3.
What is the clinical significance of the sodium nitroprusside test?
What is the basis of the Molisch test being used to differentiate aldehydes and ketones from
carbohydrates?
4.
Esters are
found in some medic ines
b. often fragrant
essential oils in fruits and flower
41.
d. found in some types of flavorings
all of the above
e.
c.
Chapter 8 Solutions
Organic Chemistry (6th Edition)
Ch. 8.1 - Problem 8.1 Label the and carbons in each alkyl...Ch. 8.2 - Problem 8.2 Classify each alkene in the following...Ch. 8.2 - Prob. 3PCh. 8.2 - Prob. 4PCh. 8.2 - Problem 8.5 Label each pair of alkenes as...Ch. 8.2 - Problem 8.6 Which alkene in each pair is more...Ch. 8.2 - Problem 8.7 Several factors can affect alkene...Ch. 8.4 - Prob. 8PCh. 8.4 - Prob. 9PCh. 8.4 - Prob. 10P
Ch. 8.4 - Prob. 11PCh. 8.5 - Problem 8.12 What alkenes are formed from each...Ch. 8.6 - Prob. 13PCh. 8.6 - Problem 8.14 What alkenes are formed from each...Ch. 8.6 - Problem 8.15 How does each of the following...Ch. 8 - 8.24 Rank the alkenes shown in the ball-and-stick...Ch. 8 - Prob. 25PCh. 8 - 8.26 What is the major E2 elimination product...Ch. 8 - Prob. 27PCh. 8 - Prob. 28PCh. 8 - Prob. 29PCh. 8 - 8.30 Label each pair of alkenes as constitutional...Ch. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Prob. 33PCh. 8 - For each of the following alkenes, draw the...Ch. 8 - Prob. 35PCh. 8 - Prob. 36PCh. 8 - Prob. 37PCh. 8 - What alkene is the major product formed from each...Ch. 8 - Prob. 39PCh. 8 - Prob. 41PCh. 8 - Draw the products formed when each dihalide is...Ch. 8 - Draw all of the substitution and elimination...Ch. 8 - Prob. 56PCh. 8 - 8.59 Draw a stepwise, detailed mechanism for each...Ch. 8 - Draw the major product formed when...Ch. 8 - Draw a stepwise, detailed mechanism for the...Ch. 8 - Explain why the reaction of with gives ...Ch. 8 - Draw a stepwise detailed mechanism that...Ch. 8 - Prob. 63PCh. 8 - 8.65 Explain the selectivity observed in the...Ch. 8 - Prob. 65PCh. 8 - Prob. 66PCh. 8 - 8.68 (a) Draw all products formed by treatment of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
During the early part of the 20th century, sulfanilamide (an antibacterial drug) was only administered by injec...
Elementary Principles of Chemical Processes, Binder Ready Version
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, & Biological Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Classify each structure according to its functional class. H A B Darrow_forwardWhich of the following is not a physical property of alcohols or phenols? A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Due to hydrogen bonding, boiling points of alcohols is much higher than those of corresponding alkanes. C. The hydroxyl group of an alcohol is nonpolar. D. Phenols are generally only slightly soluble in water.arrow_forwardEXPLAIN THE INTERACTION BETWEEN THE SOLUTE AND SOLVENT IN TERMS OF THEIR STRUCTURE. 1. Why is Ethanol soluble in water? 2. Why is Acetic Acid soluble in water? 3. WHy is Ester insoluble in water? 4. Why is Ethanal soluble in water? 5. Why is Propanol soluble in water?arrow_forward
- 5. What is the most dominant intermolecular forces of attraction that exist between: a. water and limonene, the major monoterpene component of lemon essential oil? b. water and constituent ions of sodium chloride? 6. Between the two answers in number 6, which pair exhibits a stronger attraction between the molecules? 7. Describe what happens during salting-out process in terms of the forces of attraction involved as sodium chloride is added into the water-essential oil mixture.arrow_forward19. For the structure of "Uscharidin", a poisonous natural product, consider the following statements and select the incorrect one. a. The structure has two alcohol groups b. The structure has two ketones C. The structure has a trisubstituted double bond d. The structure does not have a carboxylic acid H.C oh and. The structure has an ester OH H,C 20. The relationship that exists for the following two pairs of stereoisomers is: HC H.C CH H,C OH H,C. OH H. CH H. H. couple A couple a. both pairs are constitutional isomers b. couple A: diastereoisomers: couple B: constitutional isomers C. couple A: enantiomers; couple B: constitutional isomers. d. couple A: enantiomers; couple B: enantiomers and. None of the abovearrow_forwardFats and oils are triesters of what alcohol? A) ethanol B) isopropanol C) glycerol D) ethylene glycol E) methanolarrow_forward
- Most simple ketones and aldehydes exist mainly as the keto tautomer form. A. True B. Falsearrow_forward1. Which statement is correct? A. Aldehydes and ketones can hydrogen bond with other aldehydes and ketones. B. Aldehydes and ketones can hydrogen bond with water. C. Aldehydes and ketones cannot hydrogen bond with water. D. Aldehydes and ketones are always soluble in water.arrow_forwardRefer to the attached picture for the following questionWhat property is expected in the organic product in the reaction?A.Solubility in alcohol is increasedB.Polarity of the compound is decreasedC.A cis-configuration is expected.D.The electrophile becomes a nucleophile.arrow_forward
- Purification of Organic Compounds What type of organic compounds can be purified by distillation?arrow_forwardbe classified as what type of compound? CH3 - C CHO CH3 a Butanone mayarrow_forwardWhich of the following statements is true? a. Both aldehydes and ketones are easily oxidized.b. Neither aldehydes nor ketones are easily oxidized.c. Aldehydes are easily oxidized, but ketones are not.d. Ketones are easily oxidized, but aldehydes are not. 2. Which of the following is obtained by oxidizing hexanal? a. hexanol b. 2-hexanone c. hexanoic acid d. none of these 3. Which of the following oxidizing agents oxidizes aldehydes in the body? a. NADH b. NAD+c. O2 d. none of these 4. Which of the following will be obtained by the reaction of pentanal with H2 in the presence of a transition metal catalyst? a. pentene b. 1-pentanolc. 2-pentanol d. pentanoic acid 5. The reaction of which combination of reactants result in the formation of a hemiacetal? a. ketone and alcohol b. ketone and aldehydec. ketone and carboxylic acid d. none of thesearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning



Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY