7.18. The compressed-air-driven water rocket shown in Fig. 7.26 is ejecting water vertically downward through a frictionless nozzle. The exit area of the nozzle is 1 in². When the pressure and elevation are as shown, how much thrust does the rocket produce? Air 5 psig h 8 ft FIGURE 7.26
7.18. The compressed-air-driven water rocket shown in Fig. 7.26 is ejecting water vertically downward through a frictionless nozzle. The exit area of the nozzle is 1 in². When the pressure and elevation are as shown, how much thrust does the rocket produce? Air 5 psig h 8 ft FIGURE 7.26
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question
100%

Transcribed Image Text:7.18. The compressed-air-driven water rocket shown in Fig. 7.26 is ejecting water
vertically downward through a frictionless nozzle. The exit area of the nozzle is
1 in². When the pressure and elevation are as shown, how much thrust does the
rocket produce?
Air
5 psig
h=8 ft
FIGURE 7.26
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
hi i am not seeing your unit conversion for step 2 can u plz clarify
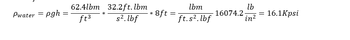
Transcribed Image Text:Pwater = pgh
=
62.4lbm 32.2ft. lbm
s².lbf
ft³
*
8ft=
=
lbm
ft.s².lbf
lb
16074.2 =
in²
16.1Kpsi
Solution
Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The