
Concept explainers
Carbon–carbon bond dissociation enthalpies have been measured for many
Without referring to Table
has the lower carbon–carbon bond-dissociation enthalpy, and explain the reason for your
choice.
Ethane or propane
Propane or
Cyclobutane or cyclopentane
Interpretation:
In each of the given pairs, the alkane having lower carbon-carbon bond dissociation enthalpy is to be identified and the reason for this is to be explained.
Concept introduction:
Species that contain unpaired electrons are called free radicals.
Alkyl radicals are described by the presence of carbon with three bonds. The alkyl radicals are classified as primary, secondary or tertiary on the basis of the number of carbon atoms directly attached to the carbon atom bearing unpaired electron.
Similar to carbocation’s, free radicals are stabilized by alkyl substituents. The stability order of alkyl radicals is
In a hemolytic cleavage, each atom of the bond keeps one of the electrons in the bond.
The bond dissociation energy represents the stability of radical formed.
Answer to Problem 16P
Solution:
a) Propane has the lower carbon-carbon bond dissociation enthalpy because it produces more stable free radicals.
b)
c)
d) Cyclopentane has the lower carbon-carbon bond dissociation enthalpy because it produces more stable free radicals.
Explanation of Solution
a) The given alkanes are ethane and propane.
The cleavage of the carbon-carbon bond in propane produces one methyl radical and one ethyl radical.
The ethyl radical is a primary radical and is more stable than the methyl radical. Hence propane produces more stable radicals than ethane upon homolytic cleavage. Lower energy is required to generate free radicals in propane. Thus, propane has a lower bond dissociation enthalpy than ethane.
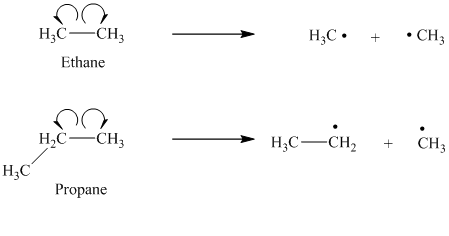
b) The given alkanes are propane and
The homolytic cleavage of the carbon-carbon bond in propane produces one methyl radical and one ethyl radical. The homolytic cleavage of the carbon-carbon bond in
The isopropyl radical is a secondary radical and is more stable than the ethyl radical, which is a primary radical. Hence
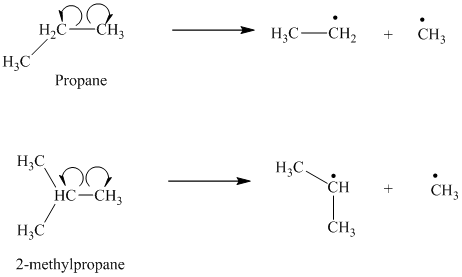
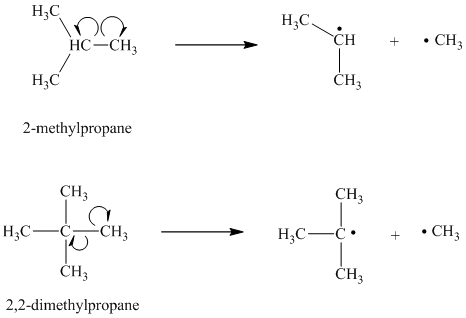
c) The given alkanes are
The homolytic cleavage of the carbon-carbon bond in
The tertiary butyl radical is a tertiary radical and is more stable than the isopropyl radical, which is a secondary radical. Hence
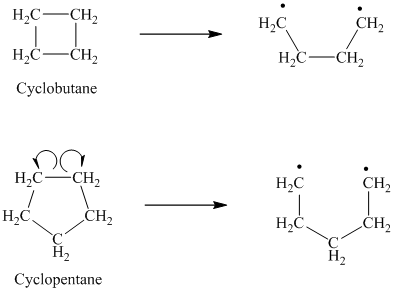
d) The given alkanes are cyclobutane and cyclopentane.
The homolytic cleavage of cyclobutane produces two radicals, which are attached to each other. Both the unpaired electrons are present on primary carbon atoms.
The homolytic cleavage of cyclopentane also produces two radicals, which are attached to each other. Both the unpaired electrons are present on primary carbon atoms.
In the radicals produced by cyclopentane, there is one alkyl substituent more as compared to the radicals in cyclobutane. The more the alkyl substituents, the more stable is the radical. Hence, in cyclopentane, the radicals are slightly more stable as compared to cyclobutane. Hence, cyclopentane has a lower bond dissociation enthalpy than cyclobutane.
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry - Standalone book
- Carbon–carbon bond dissociation enthalpies have been measured for many alkanes. Identify the alkane in each of the following pairs that has the lower carbon–carbon bond-dissociation enthalpy, and explain the reason for your choice. (a) Ethane or propane (b) Propane or 2-methylpropane (c) 2-Methylpropane or 2,2-dimethylpropane (d) Cyclobutane or cyclopentanearrow_forwardWhat is the enthalpy change to convert one mole of methane to methanol?arrow_forwardClassify alkyl halides as primary, secondary, tertiary, aliphatic, or benzenearrow_forward
- Write a balanced equation for the complete combustion of 2,3-dimethylbutane. Use the molecular formula for the alkane (C before H) and the smallest possible integer coefficients.arrow_forwardExplain the role strain plays on stability and reactivity of a cyclic alkane such as cyclopropane.arrow_forwardUsing the table of bond energies estimate the heat of combustion of one mole of 2,4-dimethyl-1,3-pentadiene.arrow_forward
- Consider the three isomeric alkanes: hexane, 2, 3-dimethylbutane, and 2-methylpentane. Which of the following correctly lists these compounds in order of increasing boiling point? The more to the right, the higher the BP.arrow_forwardDraw a structural formula for the product formed upon hydroboration/oxidation of the alkene below. CH3 • Use wedge and hash bonds ONLY for rings. • Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer.arrow_forwardAH° values obtained for a series of similar reactions are one set of experimental data used to determine the relative stability of alkenes. Explain how the following data suggest that cis-2-butene is more stable than 1-butene (Section 12.3A). CH2=CHCH,CH, + H2 CH;CH2CH2CH3 AH° =-127 kJ/mol 1-butene CH3 CH3 C=C + H2 CH3CH,CH,CHg AH° =-120 kJ/mol cis-2-butenearrow_forward
- Following are structural formulas for 1,4-dioxane and piperidine. 1,4-Dioxane is a widely used solvent for organic compounds. Piperidine is found in small amounts in black pepper (Piper nigrum). (a) Complete the Lewis structure of each compound by showing all unshared electron pairs. (b) Predict bond angles about each carbon, oxygen, and nitrogen atom. (c) Describe the most stable conformation of each ring and compare these conformations with the chair conformation of cyclohexane.arrow_forwardWhat functional group distinguishes each of the following hydrocarbon derivatives? a. halohydrocarbons b. alcohols c. ethers d. aldehydes e. ketones f. carboxylic acids g. esters h. amines Give examples of each functional group. What prefix or suffix is used to name each functional group? What are the bond angles in each? Describe the bonding in each functional group. What is the difference between a primary, secondary, and tertiary alcohol? For the functional groups in ah, when is a number required to indicate the position of the functional group? Carboxylic acids are often written as RCOOH. What does COOH indicate and what does R indicate? Aldehydes are sometimes written as RCHO. What does CHO indicate?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





