
Suppliers of radioisotopically labeled compounds usually provide each product as a mixture Of labeled and unlabeled material. Unlabeled material is added deliberately as a carrier, partly because the specific activity of the carrier-free product is too high to be useful and partly because the product is more stable at lower specific activities. Using the radioactive decay law, calculate the
following.
a. The specific activity Of carrier-free [22P]-orthophosphate, in mCi/mmol.
b. The fraction Of H atoms that are radioactive in a preparation Of uniform-label [3H]-leucine, provided at 10 mCi/mmol.
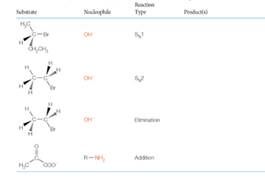
Predict the product(s) of the following reactions:
Learn your wayIncludes step-by-step video

Chapter 11 Solutions
Biochemistry: Concepts and Connections
Additional Science Textbook Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Biology: Life on Earth
Basic Chemistry (5th Edition)
Concepts of Genetics (11th Edition)
Biological Science (6th Edition)
- The net charge on the most prevalent form of bisphosphoglycerate in blood is what?arrow_forwardDehalogenase enzymes catalyze the clevage of C-X bonds. One such dehalogenase catalyzes the following reaction. An active site aspartate is thought to carry out the initial nucleophilic attack that expels the chloride. Select the most likely intermediate in the reaction. (Note the stereochemistry. Refer to P11.19 in the textbook if needed.) O O A C D OE -CH₂ H H₂C™ CO₂ H B CO₂ CI N H₂O Cr CO₂ H CO₂ нотум CH3 D CO₂ five N E CH₂arrow_forwardCellular pH is slightly alkaline (pH 7.4), explain why a seemingly slight decrease in pH to 6.8 can have very serious consequences for the organism, becoming fatal?arrow_forward
- Concentrations in biochemical systems are often very dilute. Consequently, scientific notation and logarithms are often used to express concentrations. In scientific notation, numbers are expressed as coefficient x 10" To convert a number to scientific notation, proceed as follows: 1. Move the decimal place so that there is one digit in front of the decimal. 2. Account for the moved decimal in the value of x. If the decimal moved to the right, x is negative; if it moved to the left, x is positive. A logarithm is basically an exponent. Unless otherwise indicated, a logarithm is the a of 10". The numbers after the decimal point are significant; the number before the decimal just identify the location of the decimal point for the number. Notice that it is easy to estimate a logarithm from scientific notation; it's the exponent! Logarithms are commonly used to express the concentration of H. The pH is defined as pH= log (In), where the base number is 2.303. The same general rules as logs…arrow_forwardStoichiometric equations can be used to represent the growth of microorganisms provided a 'molecular formula' for the cells is available. The molecular formula for biomass is obtained by measuring the amounts of C, N, H, O, and other elements in cells. For a particular bacterial strain, the molecular formula was determined to be C4.4H7.301.2No.86. These bacterial cells are grown under aerobic conditions with hexadecane (C16H34) as substrate. The reaction equation describing growth is: 1 C16H34 + 16.28 O2 +1.42 NH3 → 1.65 C4.4H7.301.2No.86 + 8.74 CO2+ 13.11 H20 You are in charge of a small batch fermenter for growing the bacteria you need to produce 8.2 kg of cells for inoculation of a pilot-scale reactor. a) What minimum amount of hexadecane substrate (in kg) must be contained in your culture medium? Assume 100% conversion of hexadecane to cells. b) What must be the minimum concentration of hexadecane (in kg/m3) in the medium if the fermenter working volume is 5.8 cubic meters? c) What…arrow_forwardQuinine ( C20 H24 N2 O2) is the most important alkaloid derived from cinchona bark. It is used as an antimalarial drug. For quinine, pK, = 5.1 and pK, = 9.7 ( pKp = – log Kp). Only 1 g quinine will dissolve in 1920.0 mL of solution. Calculate the pH of a saturated aqueous solution of quinine. Consider only the reaction | Q+ H2O= QH+ + OH- described by pK, where Q = quinine. pH =arrow_forward
- The four-carbon sugar erythrose-4-phosphate is the starting material in the synthesis of many compounds. Name two important examples.arrow_forwardPenicillin G is a natural antibiotic that is useful for treating infections caused by Gram positive bacteria. What is the functional benefit of the semi-synthetic antimicrobials carbenicillin and ampicillin, generated by chemical modification of the R group so their R groups each are a bit different than the R group seen with penicillin G?arrow_forwardA vial of Doxorubicin reads 0•5g per vial. Instructions say to reconstitute each 12mg with 2•5ml of NS. How many ml of NS will be needed to reconstitute the vial of the recommended concentration? please show workingarrow_forward
- The isoelectric point of eIF4a is 5.02. Students are given a sample of cell lysate containing eIF4A at pH 7.4. They are also provided with two buffers of pH 7.4 to use for ion exchange chromatography. One buffer has a very high salt concentration (1 M), the other has a low salt concentration (0.1 M). Mariela decides to use an anion exchange column, while Ashok chooses a cation exchange column. Who made the better decision? Provide a detailed explanation of why one student will end up with a purer sample of eIF4A. Which buffer will you use while washing away impurities (high or low salt), and which would you use to remove eIF4A from the column? Explain your choice.arrow_forwardCalculate the amount of rice bran (12.5% CP), corn (8.5% CP), soybean meal (49% CP) and copra meal (20% CP) in formulating 100 kg diet with 20% CP. Assume that the energy mixture will have a ratio of 2:1 and protein ingredients proportion is 1:3. Show your solution:arrow_forwardWhen a mixture of 3-phosphoglycerate and 2-phosphoglycerate is incubated at 25°C in the presence of the enzyme phosphoglycerate mutase (which catalyzes the intervconversion of the two substances) until equilibrium is reached, the final mixture contains 6 times as much 2-phosphoglycerate as 3-phosphoglycerate. Which of the following statements is most nearly correct when applied to the reaction shown below: 3-phosphoglycerate ------> 2-phosphoglycerate (R=8.315 J/mole.K) A. △G°' = +12.7 kJ/mole B. △G°' = 0 C. △G°' is incalculably large and positive D. △G°' = -4.44 kJ/mole E. △G°' cannot be calculated from the information gvenarrow_forward
