
Concept explainers
For each of the following reactions, identify the Brønsted–Lowry acids and bases:
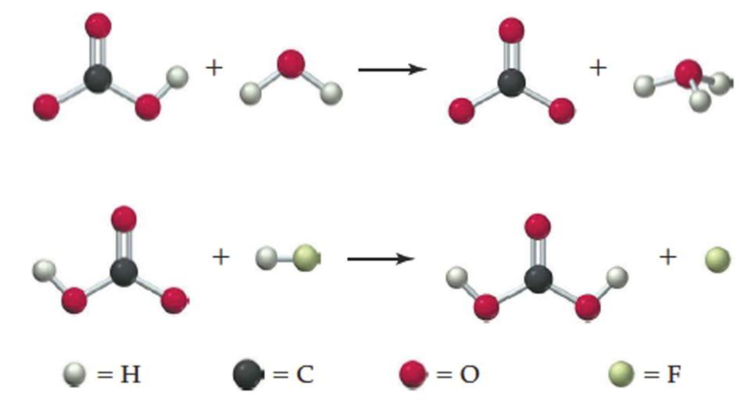
Interpretation:
For given reactions, Bronsted-Lowry acids and bases has to be identified.
Concept introduction:
Bronsted-Lowry acid and bases:
The chemical species which is able to donate hydrogen ion to other substance known as Bronsted-Lowry acid and the species which is able to accept hydrogen ion from another species known as Bronsted-Lowry base.
Answer to Problem 14.33CP
In first reaction,
In second reaction,
Explanation of Solution
Given
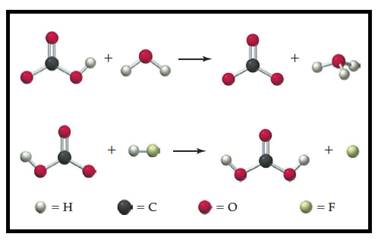
Figure 1
In first reaction:
The chemical species which is able to donate hydrogen ion to other substance known as Bronsted-Lowry acid. In first reaction
The species which is able to accept hydrogen ion from another species known as Bronsted-Lowry base. In this reaction,
In second reaction:
The chemical species which is able to donate hydrogen ion to other substance known as Bronsted-Lowry acid. In first reaction
The species which is able to accept hydrogen ion from another species known as Bronsted-Lowry base. In this reaction,
Want to see more full solutions like this?
Chapter 14 Solutions
General Chemistry: Atoms First
- Write equations that show H2PO4- acting both as an acid and as a base.arrow_forwardWrite balanced molecular equations to illustrate the following characteristic reactions of acids, using sulfuric acid (H2SO4). a. Reaction with water to form hydronium ions b. Reaction with the solid oxide CaO c. Reaction with the solid hydroxide Mg(OH)2 d. Reaction with the solid carbonate CuCO3 e. Reaction with the solid bicarbonate KHCO3 f. Reaction with Mg metalarrow_forwardWrite equations that show NH3 as both a conjugate acid and a conjugate base.arrow_forward
- Write a formula for the conjugate base formed when each of the following behaves as a Brnsted acid: a. HSO4 b. CH3NH3+ c. HClO4 d. NH4+ e. HClarrow_forwardWrite balanced molecular equations to illustrate the following characteristic reactions of acids, using nitric acid (HNO3). a. Reaction with water to form hydronium ions b. Reaction with the solid oxide CaO c. Reaction with the solid hydroxide Mg(OH)2 d. Reaction with the solid carbonate CuCO3 e. Reaction with the solid bicarbonate KHCO3 f. Reaction with Mg metalarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





