
We have made many measurements of coffee cooling in a ceramic coffee cup. We realize that as the coffee cools. It gradually reaches room temperature. Consequently, we report the value of the coffee temperature in degrees above room temperature (so after a long time, the temperature rise will be equal to 0). Also, we realize that the hotter the coffee is initially (above room temperature), the longer it will take to cool. The values presented here are in degrees Fahrenheit.
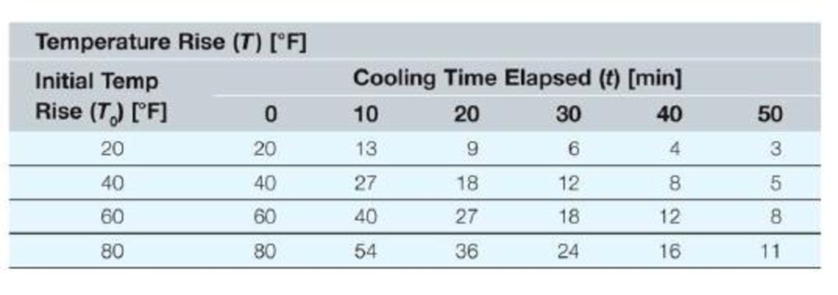
Write a MATLAB function that will perform a single interpolation given five numbers as input arguments and return the interpolated value as the only function output.
Write a MATLAB program that will calculate the following scenarios. Store each part in a different variable (e.g., part (a) should be stored in a variable named PartA, part (b) should be stored in a variable named PartB, etc.),
a. What is the temperature (rise) of the cup of coffee after 37 minutes if the initial rise of temperature is 40 degrees Fahrenheit?
b. If the coffee cools for 30 minutes and has risen 14 degrees Fahrenheit at that time, what was the initial temperature rise?
c. Find the temperature rise of the coffee at 17 minutes if the initial rise is 53 degrees Fahrenheit.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
Additional Engineering Textbook Solutions
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
Engineering Mechanics: Dynamics (14th Edition)
Mechanics of Materials
Engineering Mechanics: Statics
INTERNATIONAL EDITION---Engineering Mechanics: Statics, 14th edition (SI unit)
- Water freezes at 0° Celsius and 32° Fahrenheit and it boils at 100°C and 212°F. (a) Find a linear function F that expresses temperature in the Fahrenheit scale in terms of degrees Celsius C. Use this function to convert 5°C into Fahrenheit. °F (b) Find a linear function C that expresses temperature in the Celsius scale in terms of degrees Fahrenheit F. Use this function to convert 120°F into Celsius. °℃ (c) Is there a temperature n such that F(n) = C(n)? (If an answer does not exist, enter DNE.)arrow_forwardis a mass hanging by a spring under the influence of gravity. The force due to gravity, Fg, is acting in the negative-y direction. The dynamic variable is y. On the left, the system is shown without spring deflection. On the right, at the beginning of an experiment, the mass is pushed upward (positive-y direction) by an amount y₁. The gravitational constant g, is 9.81 m/s². DO C.D Frontly у Your tasks: No Deflection m k Fg = mg Initial Condition y m k Write down an expression for the total energy If as the sum Write down an expression for the total energy H Fg = mg Figure 3: System schematic for Problem 4. Yi & X Write down, in terms of the variables given, the total potential energy stored in the system when it is held in the initial condition, relative to the system with no deflection. as the sum of potential and kinetic energy in terms of y, y, yi C After the system is released, it will start to move. Write down an expression for the kinetic energy of the system, T, in terms of…arrow_forwardYou have the six pieces of metal listed below plus a beaker of water containing 400. g of water. The water temperature is 21.0 °C. Metals Specific Heat (J/g. K) Mass (g) 1. Al 0.9002 100. 2. Al 3. Au 4. Au 5. Zn 6. Zn 0.9002 0.1289 0.1289 0.3860 0.3860 50. 100. 50. 100. 50. The specific heat capacity of water is 4.184 J/g °C.arrow_forward
- The specific heat of copper is 0.093 cal/(g.°C) and the specific heat of gold is 0.031 cal/(g.°C). If 5.8 cal is supplied to one gram of copper and one gram of gold, the RATIO of temperature increase of gold to that of copper isarrow_forwardConsider a process to prepare a metal for a certain application. There are five parameters that must be considered: temperature, quenching rate, cooling time, carbon content, CO₂ concentration. It is desired to determine which of these parameters has the most influence on the process. There are two levels for each parameter as shown below. Temperature (°C) Quenching Rate(°C/s) Cooling time (s) Carbon Content (wt% C) CO₂ Concentration (%) Eight experiments were defined as follows: Experiment Carbon Content (wt% C) 1 2 3 4 5 6 7 8 1) What size orthogonal array should be used for evaluation (assume noise is negligible)? 2) Generate the array with the level values (i.e. Level 1 and Level 2). 1 1 1 1 6 6 6 6 Quenching Rate (°C/s) 35 35 140 140 35 35 140 140 Four trials were run for the experiments defined above: Experiment 1 2 3 4 5 6 7 8 Level 1 760 35 1 1 5 T1 68.00 69.84 74.36 71.71 91.27 54.39 64.65 60.31 Cooling Time 1 1 300 300 300 300 1 1 T2 61.41 64.76 61.30 58.42 90.89 Level 2 900…arrow_forwardConsider an introductory thermodynamics class experiment used to demonstrate phase change phenomena. A beaker of water is heated, and its temperature measured over time to establish the temperature at which boiling occurs. The results, shown in the Figure below, are for three separate tests conducted on different days by different student groups using the same equipment and method. Why might the data from three seemingly identical tests show different results? Temperature (°C) 101 100 99 98 97 96 95 94 1 2 3 4 Time (min) 5 Boiling region 100.3 100.1 99.8 Boiling point results Test 1 (762 mm Hg) Test 2 (754 mm Hg) Test 3 (767 mm Hg) 6 7arrow_forward
- Jes 1:01 i docs.google.com/forms what is the Physicists recognize four fundamental forces.* Flectrical force O - Gravitational force -Strong nuclear force Weak nuclear force O -Muscular force Force of friction Ff is described by • Ff = µN * O p synovlal fluld in the Joints P -coefficient of fraction between two surface in The temperature of the human body is normally about 98.6°F.calculate the temperature of the body "C* oC = (5/9)x(oF-32) =5/9(98.6- 32)-37 oc oc=(9/5)x(of -32) =9/5(98,6-32) =13,3 Under resting conditions the body energy is being used as follows * O 27% by the liver and splee O 20-%by the skeletal musules Under resting conditions the body energy is being used as follows. O 19% by the brain. 15% by the kidney. We can write the first law of thermodynamics as: * O AU-AQ -A.. O AU-AQ +AW. 1kcal = J O 1 Kcal =4184j. O O O Oarrow_forwardCalculate the density, weight density, specific volume of 2 liters of a fluid of 5 kg mass (ENTER ONLY THE VALUES IN THE BOXES BY REFERRING THE UNIT GIVEN IN BRACKETS) density of fluid (unit in kg/m³) = One possible correct answer is: 2500 weight density of fluid is (Unit in N/m³) =- One possible correct answer is: 24525 specific volume (unit in mm3/kg) : %3D One possible correct answer is: 400000 specific gravity One possible correct answer is: 2.5arrow_forwardQ8): To find how much heat is required to bring a kettle of water to its boiling point, you are asked to calculate the specific heat of water at 61°C. The specific heat of water is given as a function of time in Table below. Temperature, T Specific heat, C₂ (°C) J kg-°C 22 42 52 82 100 4181 4179 4186 4199 4217 Determine the value of the specific heat at 7=61°C using the direct T method of interpolation and a third order polynomial. Find the absolute relative approximate error for the third order polynomial approximation (Lagrange Method).arrow_forward
- is a mass hanging by a spring under the influence of gravity. The force due to gravity, Fg, is acting in the negative-y direction. The dynamic variable is y. On the left, the system is shown without spring deflection. On the right, at the beginning of an experiment, the mass is pushed upward (positive-y direction) by an amount y₁. The gravitational constant g, is 9.81 m/s². No Deflection m k Fg = mg Initial Condition m k Fg = mg Figure 3: System schematic for Problem 4. Yi 8 Your tasks: A Write down, in terms of the variables given, the total potential energy stored in the system when it is held in the initial condition, relative to the system with no deflection. B Write down an expression for the total energy H as the sum of potential and kinetic energy in terms of y, y, yi and element parameters. Will H change as the mass moves? C After the system is released, it will start to move. Write down an expression for the kinetic energy of the system, T, in terms of position, y, the initial…arrow_forwardre | https://eagleonline.hccs.edu/courses/156500/quizzes/969413/take Question 39 5 pts Perform the following temperature unit conversions: (a) Convert a temperature of 52°C to 'F °R, and K. (b) Convert a temperature of -40°F to °C, K, and °R. Relevant formulae are given below T (°C) = T (K) - 273.15 T ('F) = T (°R) - 459.67 T(F) = 1.8T (°C) + 32 Upload Choose a File 5 pts tion 40arrow_forwardThe below shown graph is drawn from the tabulated values of steel which we measured during the experiment of thermal conductivity: (Consider the value of heater power (Q') and the area of cross section (A) of the material from the tabulated values) X-axis 1 unit = 1 cm y-axis 1 unit = 10°C 80- 70- 60- 50- 40- 30- 20- 10- 6. Distance X in cm 4. 5. 8. 10 Temperature T degree Celsiusarrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
