
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN: 9781133104261
Author: Raymond A. Serway, John W. Jewett
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17.6, Problem 17.5QQ
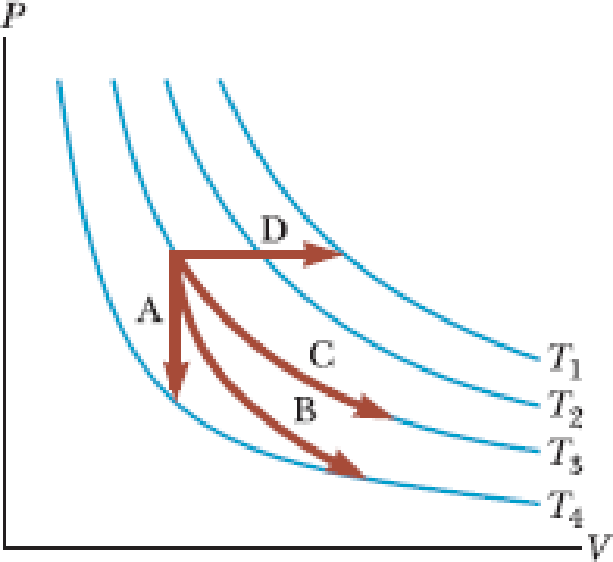
Characterize the paths in Figure 17.10 as isobaric, isovolumetric, isothermal, or adiabatic. For path B, Q = 0. The blue curves are isotherms.
Figure 17.10 (Quick Quiz 17.5)
Identify the nature of paths A, B, C, and D.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
0.5 moles of cinnamaldehyde gas that has an initial volume of ten liters expands under the following conditions: 185°F and 800mmHg external pressure.
a. How much work is done? Express your answer in L atm.
b. Assuming that the container is cylindrical with a base radius of 10cm and can only expand
vertically, how much kinetic energy does a single molecule possesses if it travels from base
to base of the expanded cylinder in 10us? Express your answer in J
I just need help with part D
Problem 6: There are lots of examples of ideal gases in the universe, and they exist in many different conditions. In this problem we will examine what the temperature of these various phenomena are.
Part (a) Give an expression for the temperature of an ideal gas in terms of pressure P, particle density per unit volume ρ, and fundamental constants.
Answer: T = P/( ρ kB )
Part (b) Near the surface of Venus, its atmosphere has a pressure fv= 96 times the pressure of Earth's atmosphere, and a particle density of around ρv = 0.92 × 1027 m-3. What is the temperature of Venus' atmosphere (in C) near the surface?
Answer: Tv = 490.55
Part (c) The Orion nebula is one of the brightest diffuse nebulae in the sky (look for it in the winter, just below the three bright stars in Orion's belt). It is a very complicated mess of gas, dust, young star systems, and brown dwarfs, but let's estimate its temperature if we assume it is a uniform ideal gas. Assume it is a…
Instructions
Answer the following questions.
ⒸPV Diagrams
Which of the following PV diagrams have at least one step that is ISOBARIC and at least one step that is ISOVOLUMETRIC? Choose all that apply.
X
P
V
P
✓
✔
P
V
P
V
✔
P
V
P
D
V
[]
Chapter 17 Solutions
Principles of Physics: A Calculus-Based Text
Ch. 17.2 - Prob. 17.1QQCh. 17.3 - Prob. 17.2QQCh. 17.3 - Prob. 17.3QQCh. 17.5 - Prob. 17.4QQCh. 17.6 - Characterize the paths in Figure 17.10 as...Ch. 17.7 - (i) How does the internal energy of an ideal gas...Ch. 17.10 - Prob. 17.7QQCh. 17 - Prob. 1OQCh. 17 - A 100-g piece of copper, initially at 95.0C, is...Ch. 17 - Prob. 3OQ
Ch. 17 - Prob. 4OQCh. 17 - Prob. 5OQCh. 17 - Prob. 6OQCh. 17 - Prob. 7OQCh. 17 - Prob. 8OQCh. 17 - Prob. 9OQCh. 17 - Prob. 10OQCh. 17 - Star A has twice the radius and twice the absolute...Ch. 17 - If a gas is compressed isothermally, which of the...Ch. 17 - When a gas undergoes an adiabatic expansion, which...Ch. 17 - Ethyl alcohol has about one-half the specific heat...Ch. 17 - Prob. 15OQCh. 17 - Prob. 1CQCh. 17 - Prob. 2CQCh. 17 - Pioneers stored fruits and vegetables in...Ch. 17 - Why is a person able to remove a piece of dry...Ch. 17 - Prob. 5CQCh. 17 - Prob. 6CQCh. 17 - It is the morning of a day that will become hot....Ch. 17 - You need to pick up a very hot cooking pot in your...Ch. 17 - Rub the palm of your hand on a metal surface for...Ch. 17 - Prob. 10CQCh. 17 - Prob. 11CQCh. 17 - Prob. 12CQCh. 17 - On his honeymoon, James Joule traveled from...Ch. 17 - Consider Joules apparatus described in Figure...Ch. 17 - Prob. 3PCh. 17 - Prob. 4PCh. 17 - Prob. 5PCh. 17 - Prob. 6PCh. 17 - Prob. 7PCh. 17 - Prob. 8PCh. 17 - Prob. 9PCh. 17 - Prob. 10PCh. 17 - Prob. 11PCh. 17 - Prob. 12PCh. 17 - Prob. 13PCh. 17 - Prob. 14PCh. 17 - In an insulated vessel, 250 g of ice at 0C is...Ch. 17 - Prob. 16PCh. 17 - Prob. 17PCh. 17 - Prob. 18PCh. 17 - A 1.00-kg block of copper at 20.0C is dropped into...Ch. 17 - A resting adult of average size converts chemical...Ch. 17 - Prob. 21PCh. 17 - Prob. 22PCh. 17 - An ideal gas is enclosed in a cylinder with a...Ch. 17 - Prob. 24PCh. 17 - Prob. 25PCh. 17 - A sample of an ideal gas goes through the process...Ch. 17 - A thermodynamic system undergoes a process in...Ch. 17 - A gas is taken through the cyclic process...Ch. 17 - Consider the cyclic process depicted in Figure...Ch. 17 - Why is the following situation impossible? An...Ch. 17 - An ideal gas initially at 300 K undergoes an...Ch. 17 - In Figure P17.32, the change in internal energy of...Ch. 17 - Prob. 33PCh. 17 - Prob. 34PCh. 17 - Prob. 35PCh. 17 - Prob. 36PCh. 17 - Prob. 37PCh. 17 - One mole of an ideal gas does 3 000 J of work on...Ch. 17 - A 1.00-mol sample of hydrogen gas is heated at...Ch. 17 - A sample of a diatomic ideal gas has pressure P...Ch. 17 - Prob. 41PCh. 17 - Prob. 42PCh. 17 - Prob. 43PCh. 17 - Review. This problem is a continuation of Problem...Ch. 17 - Prob. 45PCh. 17 - A 2.00-mol sample of a diatomic ideal gas expands...Ch. 17 - Prob. 47PCh. 17 - An ideal gas with specific heat ratio confined to...Ch. 17 - Prob. 49PCh. 17 - Prob. 50PCh. 17 - Prob. 51PCh. 17 - Prob. 52PCh. 17 - Air (a diatomic ideal gas) at 27.0C and...Ch. 17 - Prob. 54PCh. 17 - Prob. 55PCh. 17 - Prob. 56PCh. 17 - Prob. 57PCh. 17 - Prob. 58PCh. 17 - Prob. 59PCh. 17 - Prob. 60PCh. 17 - Prob. 61PCh. 17 - Prob. 62PCh. 17 - The surface of the Sun has a temperature of about...Ch. 17 - Prob. 64PCh. 17 - At high noon, the Sun delivers 1 000 W to each...Ch. 17 - A theoretical atmospheric lapse rate. Section 16.7...Ch. 17 - Prob. 67PCh. 17 - A sample of a monatomic ideal gas occupies 5.00 L...Ch. 17 - An aluminum rod 0.500 m in length and with a...Ch. 17 - Prob. 70PCh. 17 - Prob. 71PCh. 17 - Prob. 72PCh. 17 - Prob. 73PCh. 17 - Prob. 74PCh. 17 - Prob. 75PCh. 17 - Prob. 76PCh. 17 - Prob. 77PCh. 17 - Prob. 78PCh. 17 - Prob. 79PCh. 17 - Prob. 81PCh. 17 - Prob. 82PCh. 17 - Prob. 84PCh. 17 - Prob. 85PCh. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - Water in an electric teakettle is boiling. The...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Lead has a density of 11.3 103 kg/m3 at 0C. (a) What is the density of lead at 90C? (b) Based on your answer to part (a), now consider a situation in which you plan to invest in a gold bar. Would you be better off buying it on a warm day? Explain.arrow_forwardMy physics class has turned to online due to the COVID-19 and I am having trouble with some of the word problems my teacher gave me to work on. She presented me with: I am contemplating a career change. If I had 500g of stolen gold jewelry, how much energy would I have to add to it to melt it (so that it was not longer recognizable)? Pretend that it is pure gold. Would you be able to show me how to accomplish this problem so I can see the steps? Thank you for your time. John Paytonarrow_forwardFigure I shows the P-V graph for a sample of a gas at 298 K. If this sample were heated to 312 K, this would be represented by a point 25. atm Figure 1 100 V/e 50 A At the top, left end of the line Off the line, above and to the right of it. B At the lower, right end of the line D Off the line, below and to the right of it.arrow_forward
- λ=kT/21/2σp is the equation of mean free path. What is the relation of mean free path to temperature, pressure, and molecular cross section. Sketch a plot of the relationship between mean free path and the molecular cross section. Label your axes.arrow_forward0.0042 mol of gas undergoes the process shown in (Figure 1). You may want to review (Pages 503 - 508). igure p (atm) 3 2 1 0- 0 100 2 200 300 1 of 1 V (cm³) Part B What is the initial temperature in °C? Express your answer using two significant figures. T₁ = Submit Part C Tf = What is the final temperature in °C? Express your answer using two significant figures. Submit VG ΑΣΦ Provide Feedback Request Answer ΑΣΦ Request Answer ? 11? °Carrow_forwardActivity 2 Schmedrick takes his dune buggy to the gas station and fills it up to the very brim. His tank is a steel cylinder of radius 23 cm and height 45 cm (big enough to hold about 20 gallons). He burns a liter of gas getting to the beach, where both the tank and the gas heat up by 20 ºC. Both the tank and the gas expand. For steel α = 1.1 · 10-5 / ºC. For gasoline β = 9.6 · 10-4 / ºC. Does the tank overflow? Use the linear expansion formula to calculate the increase in radius of the tank: Use the linear expansion formula to calculate the increase in height of the tank: For a cylinder, V = π r 2 h. Calculate the increase in volume of the tank: Calculate the volume of gasoline at the beach before expansion. (1 cm 3 = 1 mL): Use the volume expansion formula to calculate the increase in volume of the gasoline: Conclusion:arrow_forward
- Determine the energy stored (in units of mJ) by C4 when C1 = 20 µF, C2 10 uF, C3 = 14 pF, C4 = 30 pF, and Vo = 61 V. Select one: OA. 0.93 O B. 3.97 OC. 5.95 OD. 0.65 OE. 58.05arrow_forwardThe following data (figure) describe the diffusion of a substitutional impurity atom in a polycrystalline solid. Each data set below (A, B, and C) is associated with a characteristic diffusion environment for the impurity atom. The possible diffusion environments are: surface diffusion, volume diffusion, grain boundary diffusion. Based on your understanding of solid-state diffusion mechanisms and processes, name the diffusion environment most likely associated with each data set:arrow_forwardGiven the information appearing in Fig. 4.127, determine: b.VE a. Ic. Vcc C. V e. V B d. V CE f. R₁. Vcc 20 μα | 8.2 ΚΩ '2.7 ΚΩ le + VCE • 1.2 ΚΩ FIG. 4.127 10.6 V B = 100 VEarrow_forward
- A particle gas consists of N monatomic particles each of mass m all contained in a volume V at temperature T. Your answers should be written in terms of the Boltzmann constant kB and Avogadro's number NA rather than R = NAKB- ▾ Part A Find (²), the average speed squared for each particle. Express the average speed squared in terms of the gas temperature T and any other given quantities. ▸ View Available Hint(s) (02) = Ο ΑΣΦ ? Submit ▼Part B Find U, the internal energy of the gas. Express the internal energy in terms of the gas temperature T and any other given quantities. ▸ View Available Hint(s) U = ΜΕ ΑΣΦ ? Submit Review | Constantarrow_forward1 cal = 4.184 J, specific heat of iron = 0.45 J/g°C 1. If 2087.5 kcal of chemical energy is converted to mechanical energy and then electrical energy, how many kcal and joules of electrical energy is available? Explain. 2. a) Define the term, state function. ( one of the wo assmptions made for this model, (6 (amiog o) sbam od anoizulonoo ntw bas bovoado o Jominoqo brohadut eom3 sdinoesC b) Give THREE examples of state functions. (arrow_forwarda- A box of gas has 17 distinguishable particles and 168 cells. Calculate the number of microstates. State your answer in exponential form. b. Choose the correct statements that are related to the video example of a box with a movable partition. a. Maximizing Ωtotal determines the macrostate with the maximum number of microstates. b. Maximizing Ωtotal determines the macrostate with the highest probability. c. Maximizing Ωtotal determines the macrostate that is the equilibrium state. d. Maximizing Ωtotal determines the macrostate which maximizes the number of microstates in the left half of the box.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY