
Concept explainers
- The numbers of orbiting electrons in aluminum and silver are 13 and 47, respectively. Draw the electronic configuration for each, and discuss briefly why each is a good conductor.
- Using the Internet, find the atomic structure of gold and explain why it is an excellent conductor of electricity.
(a)
To draw:
The electronic configuration of aluminum and silver. Also, state the reason due to which both aluminum and copper are considered a good conductor.
Explanation of Solution
Given:
The numbers of orbiting electrons in aluminum and silver are
The arrangement of electron inside the atom and outside the nucleus follows a particular pattern. There is a shell structure in which all electron falls and the energy of the electrons decide which shell to fall. The number of electrons in each shell is always
In shell
In Shell
Inside shell, actually electron falls in sub-shell. The name of sub-shell are
In a nutshell, the larger the value of
The electronic configuration of any element is based on below pattern:
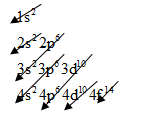
For aluminum, we know that the numbers of orbital electrons are 13.
So, arrange the electrons will be as below
The electronic configuration of Aluminum is
Here we can see, Aluminum has one free electron in its valence shell. Therefore, this free electron can move easily and hence, this causes the higher conductivity for Aluminum.
For silver, we know that the numbers of orbital electrons are 47.
So, arrange the electrons will be as follows:
The electronic configuration of silver is
Silver is a good conductor because it has free electrons in its valence shell. It is clearly seen from the electronic configuration of silver (
(b)
The atomic structure of the gold. Also, state the reason due is an excellent conductor of electricity.
Explanation of Solution
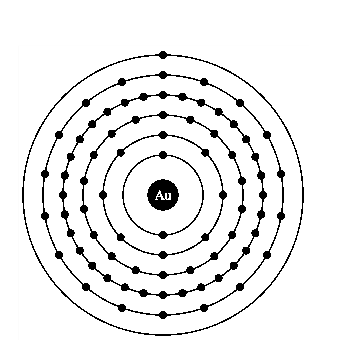
Atomic structure of gold.
The electronic configuration of any element is based on below pattern:
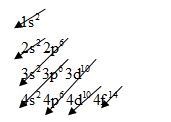
For gold, we know that the numbers of orbital electrons are
So, arrange the electrons for gold will be as follows:
The electronic configuration of gold is
Gold is a good conductor of electricity because it has free electrons in its valence shell.
It is clearly seen from the electronic configuration of gold that, s orbit is holding only one electron and maximum limit is up to
Want to see more full solutions like this?
Chapter 2 Solutions
Introductory Circuit Analysis (13th Edition)
Additional Engineering Textbook Solutions
Fundamentals of Applied Electromagnetics (7th Edition)
Electrical Engineering: Principles & Applications (7th Edition)
C How to Program (8th Edition)
Programming in C
Introduction to Java Programming and Data Structures, Comprehensive Version (11th Edition)
Web Development and Design Foundations with HTML5 (8th Edition)
- What is a proton? Where does it normally exist in an atom, and what is its charge?arrow_forwardHow many valence electrons are generally contained in materials used for insulators?arrow_forwardUsing the idea of energy bands in solid materials, explain the characteristics of the followingmaterialsa) conductorsb) insulatorsc) semiconductorsarrow_forward
- A piece of silver wire has a resistance of 1 ohm. Determine the resistance of alead wire one-third the length and one-third the diameter if the resistivity of lead wire is 30 times that of silver. NOTE: Please explain where did you get the variables so I can understand them. Thank youarrow_forwardA sample of wire (1 mm in diameter by 1 m in diameter) length) of an aluminum alloy (containing 1.2% Mn) is placed in an electrical circuit, as shown in the figure next to. A voltage drop of 432 mV is measured at wire length when it carries a 10 A current. Calculate the conductivity of this alloy.arrow_forwardP1.15. A copper wire has a diameter of 2.05 mm and carries a current of 15 A due solely to clectrons. (These values are common in residential wiring.) Each electron has a charge of -1.60 × 10-19 C. Assume that the free-electron (these are the electrons capable of moving through the copper) concentration in copper is 1029 electrons/m'. Find the average velocity of the electrons in the wire.arrow_forward
- An 18-gauge copper wire with a diameter of 1-mm carries a constant current of 1-A. The charge concentration in the wire is 8.5 x 1028 per cubic meter. Determine the electric field in mN/C. Use q = 1.602 x 10-19 and resistivity equal to 1.72x10-8 ohm-m.arrow_forwardSubject:electronic engineering Why a semiconductor acts as an insulator at 0°k and why its conductivity increases with increasing the temperature?arrow_forwardA typical Cu wire might have 2 X 1021 free electrons in 1 cm of its length. Suppose that the drift velocity of the electrons along the wire is 0.05 cm/s. (A) How many electrons would pass through a given cross section of the wire each second? (B) How large a current would be flowing in the wire? outz mduz to elob beluberlba ol uoy wollo liw ainf.nokaimdu2 to etob beluberoz edt elin M? niy ina lonaarrow_forward
- (b) Consider the diagram of a conducting wire of Figure Q1(b)6). i. Determine the charge of 7.573 x 1017 electrons. ii. If this number of electrons moves uniformly from the left end of the wire to the right end in 1 ms (millisecond), what is the current flows through the wire? iii. How many electrons must pass a given point in 1 minute to produce a current of 10 A? iv. If the charge profile across the cross section of the conductor from left to right is given by g(t) = t + 0.2est C for t2 0 and zero for t < 0, plot the profile of the current that flows across the boundary. In what direction would the current flow? v. Repeat part (iv) for the charge profile (in Coulomb) sketched in Figure Q1(b)(ii).arrow_forwardDiscuss the variation of resistivity with temperature for insulators, semiconductors and conductors. Support your discussion with resistivity versus temperature graph/curvesarrow_forwardA p-type and n-type semiconductor participates in the positive and negative charge respectively. Please choose one: A) false B) truearrow_forward
 Delmar's Standard Textbook Of ElectricityElectrical EngineeringISBN:9781337900348Author:Stephen L. HermanPublisher:Cengage Learning
Delmar's Standard Textbook Of ElectricityElectrical EngineeringISBN:9781337900348Author:Stephen L. HermanPublisher:Cengage Learning Electricity for Refrigeration, Heating, and Air C...Mechanical EngineeringISBN:9781337399128Author:Russell E. SmithPublisher:Cengage Learning
Electricity for Refrigeration, Heating, and Air C...Mechanical EngineeringISBN:9781337399128Author:Russell E. SmithPublisher:Cengage Learning

