
Concept explainers
Practice Problem ATTEMPT
ATTEMPT
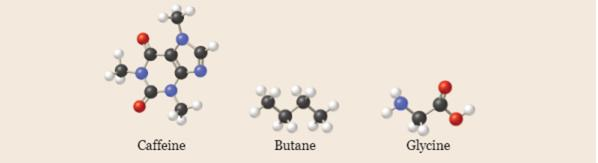
Write empirical formulas for the following molecules: (a) caffeine
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemistry
- Give the empirical formula of each of the following compounds if a sample contains the following quantities of each element. (Type your answer using the format co2 for CO2.) (a) 0.0334 mol C, 0.1000 mol H, and 0.0167 mol o (b) 16.8 g C, 1.20 g H, and 6.40 g o (c) 54.5% C, 9.2% H, and 36.3% O by massarrow_forwardPolymers are large molecules composed of simple units repeated many times. Thus, they often have relatively simple empirical formulas. Calculate the empirical formulas of the following polymers:(a) Lucite (Plexiglas); 59.9% C, 8.06% H, 32.0% O(b) Saran; 24.8% C, 2.0% H, 73.1% Cl(c) polyethylene; 86% C, 14% H(d) polystyrene; 92.3% C, 7.7% H(e) Orlon; 67.9% C, 5.70% H, 26.4% Narrow_forwardDetermine the number of moles of the compound and determine the number of moles of each type of atom in each of the following:(a) 2.12 g of potassium bromide, KBr(b) 0.1488 g of phosphoric acid, H3PO4(c) 23 kg of calcium carbonate, CaCO3(d) 78.452 g of aluminum sulfate, Al2(SO4)3(e) 0.1250 mg of caffeine, C8H10N4O2arrow_forward
- Ball-and-stick representations of benzene, a colorless liquidoften used in organic chemistry reactions, and acetylene, a gasused as a fuel for high-temperature welding, are shown below.(a) Determine the molecular formula of each. (b) Determinethe empirical formula of each.arrow_forwardQUESTION 1. N,03(g) + 6H,(g) → 2NH3(g) + 3H,0(g) how many moles of NH3(g) could be formed from the reaction of 0.22 mol of N,0, (g) with 0.87 mol of H2(g)? (AJ 0.29 mol (B) 0.44 mol (C) 0.73 mol (D) 1.1 mol (n)arrow_forwardGive the empirical formula of each of the following compounds if a sample contains the following quantities of each element. (Type your answer using the format CO2 for CO2.) (a) 0.0195 mol C, 0.039 mol H, and 0.013 mol O (b) 11.66 g iron and 5.010 g oxygen (c) 25.9% nitrogen and 74.1% oxygen by massarrow_forward
- A compound is decomposed in the laboratory and produces 1.40 g N and 0.20 g H. What is the empirical formula of the compound? (a) NΗ (c) NH2 (b) N¿H ( d) N/Ηarrow_forwardFind the empirical formula of the following compounds:(a) 0.039 mol of iron atoms combined with 0.052 mol of oxygenatoms(b) 0.903 g of phosphorus combined with 6.99 g of bromine(c) A hydrocarbon with 79.9 mass % carbonarrow_forward(1) How many moles of oxygen atoms are there in one mole of ozone molecules (O3)? (a) 6.022 x 10 23 moles (c) 1 mole (b) 2 moles (e) none of these (d) 3 molesarrow_forward
- Determine the empirical and molecular formulas of eachof the following substances:(a) Ibuprofen, a headache remedy, contains 75.69% C,8.80% H, and 15.51% O by mass and has a molar massof 206 g/mol.(b) Cadaverine, a foul-smelling substance produced bythe action of bacteria on meat, contains 58.55% C,13.81% H, and 27.40% N by mass; its molar mass is102.2 g/mol.(c) Epinephrine (adrenaline), a hormone secreted into thebloodstream in times of danger or stress, contains 59.0%C, 7.1% H, 26.2% O, and 7.7% N by mass; its molar massis about 180 amu.arrow_forwardRead each of the following statements very carefully. Then decide if the statement is CORRECT or WRONG. If the statement is WRONG, EXPLAIN why you think the statement is WRONG. (a) When the fire alarm rings continuously, leave the building and go to wait at an assembly point (b) Br has 45 neutrons (c) The molecular formula of sodium chloride is NaCl (d) The empirical formula of oxalic acid is H2C2O4 (e) Ammonium sulfide is a binary ionic compound (f) Gases condense at negative temperature on the Kelvin scale (g) A chemical reaction which produces a poisonous gas must be carried out in the fume hood (h) When your house is on fire you must call the police immediately and give them the location of your house (i) Halogens are reducing agents when they react with alkali metals (i) One of the chemical properties of sugar is its high solubility in waterarrow_forwardDetermine the simplest formulas of the following compounds: (c) nicotine, which has the composition 74.0% C, 8.65% H, and 17.4% Narrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





