
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 31.1, Problem 1P
Order the following monomers with respect to their expected reactivity toward cationic
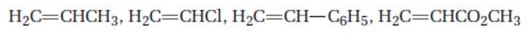
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
List the following group of monomers in order of decreasing ability to undergo cationic polymerization.
A3
Anionic polymerization of styrene is generally known to produce polymers with a narrow molecular weight distribution. Explain the reason for this.
A boron-siloxane cross linked polymer was formed from the reaction between dichlorodimethylsilane in diethyl ether with deionised water. Which was neutralised with sodium bicarbonate solution before being turned into a polymer upon the addition of boric acid. What is the structure of the polymer synthesised and what is the molecular weight of one monomer unit of this boron-siloxane cross linked polymer?
Chapter 31 Solutions
Organic Chemistry
Ch. 31.1 - Order the following monomers with respect to their...Ch. 31.1 - Order the following monomers with respect to their...Ch. 31.1 - Prob. 3PCh. 31.2 - Prob. 4PCh. 31.2 - Prob. 5PCh. 31.3 - Prob. 6PCh. 31.3 - Prob. 7PCh. 31.4 - Prob. 8PCh. 31.4 - Show the mechanism of the nucleophilic addition...Ch. 31.5 - Prob. 10P
Ch. 31.6 - Prob. 11PCh. 31.6 - Prob. 12PCh. 31.SE - Prob. 13VCCh. 31.SE - Prob. 14VCCh. 31.SE - Prob. 15MPCh. 31.SE - The polyurethane foam used for home insulation...Ch. 31.SE - Prob. 17MPCh. 31.SE - Prob. 18MPCh. 31.SE - Prob. 19MPCh. 31.SE - Identify the monomer units from which each of the...Ch. 31.SE - Prob. 21APCh. 31.SE - Draw the structure of Kodel, a polyester prepared...Ch. 31.SE - Show the structure of the polymer that results...Ch. 31.SE - Prob. 24APCh. 31.SE - Prob. 25APCh. 31.SE - 1, 3-Cyclopentadiene undergoes thermal...Ch. 31.SE - Prob. 27APCh. 31.SE - Prob. 28APCh. 31.SE - Prob. 29APCh. 31.SE - Prob. 30APCh. 31.SE - Prob. 31APCh. 31.SE - Prob. 32APCh. 31.SE - Prob. 33APCh. 31.SE - Prob. 34APCh. 31.SE - Prob. 35AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Order the following monomers with respect to their expected reactivity toward anionic polymerization, and explain your answer:arrow_forwardGrubb's catalyst ROMP (d) Demonstrate that the polymerization of propene with a simple Cp2ZrCl₂ catalyst would give rise to atactic polypropylenearrow_forwardWhen certain cycloalkenes are used in metathesis reactions, ringopening metathesis polymerization (ROMP) occurs to form a highmolecular- weight polymer, as shown with cyclopentene as the starting material. The reaction is driven to completion by relief of strain in the cycloalkene.What products are formed by ring-opening metathesis polymerization of each alkene?arrow_forward
- Chain branching is not as common with anionic polymerization as it is with free-radical polymerization and cationic polymerization. Compare the relative stabilities of the intermediates in this mechanism with those you drew for chain branching in the cationic polymerization of styrene. Explain why chain branching is less common in this anionic polymerization.arrow_forwardRank the following groups of monomers from most able to least able to undergo cationic polymerization:arrow_forward(a) Compare the characteristic features of polymerization by step-growth and chain- growth mechanisms. (b) Many alkene monomers (olefins) are readily polymerized by a free-radical mechanism. However, isobutylene is usually polymerized by a cationic mechanism. Explain.arrow_forward
- Polyester is formed by a condensation reaction between maleic anhydride and butylene glycol. Styrene is then added and polymerized under free radical polymerization condition. Describe the chemical composition and molecular architecture of the final resulting polymer. What would be the effect if maleic anhydride were replaced with adipic acid? Structures of the reacting molecules are given below. Support your answers with chemical questions (Mechanism is not required). Maleic anhydride: ,Butylene glycol: HO CH Styrene: Adipic acid: но.arrow_forwardIdentify which is suitable for radical, cataionic, anaionic, condensation polymerization, and give the resulting polymeric structure I st & NC OMearrow_forwardWhich reaction—dehydration synthesis or hydrolysis—converts a polymer to its monomers? Which one convertsmonomers to a polymer? Explain your answerarrow_forward
- One common type of cation exchange resin is prepared by polymerization of a mixture containing styrene and 1,4-divinylbenzene . The polymer is then treated with concentrated sulfuric acid to sulfonate a majority of the aromatic rings in the polymer. Q.) Show the product of sulfonation of each benzene ring.arrow_forward5. Predict the structure of the linear polymer formed by Ziegler- Natta polymerization of: (a) 2,6-dimethyl-1,6-heptadiene (b) 1,5-cyclooctadienearrow_forwardNetwork phenol-formaldehyde; linear and heavily crosslinked cis-isoprene State whether it is possible to determine whether one polymer is more likely to crystallize than the other. If it is possible, note which is the more likely and then cite reason(s) for your choice. If it is not possible to decide, state why.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY