
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.10C, Problem 5.19P
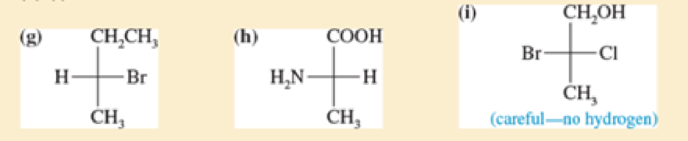
For each Fischer projection, label each asymmetric carbon atom as (R) or (S) (a)-(f) the structures in Problem5-18
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
H-
CO₂H
CH3
F
Select the appropriate stereochemical designation based on the Fischer projection shown above.
Of the four structures shown here in Fischer
projections, indicate the relationships between (a)
and (b), and between (c) and (d)?
CHO
CHO
нтон нотн
CH₂OH
(a)
CH₂OH
(b)
ÇO₂H
H-OH
-OH
-он но-
HO-
CO₂H
(c)
II
HO-H
ÇO₂H
of
A. Both pairs are enantiomers
O B. Both pairs are identical
C. (a) and (b) are enantiomers, (c) and (d)
are identical
D. (a) and (b) are identical, (c) and (d) are
enantiomers
II
CO₂H
(d)
Department
Final EXd
Q1: Coibacin B is a natural product that exhibits potent anti-inflammatory activity and potential activity in the
treatment of leishmaniasis, a disease caused by certain parasites.
HAH
H
(a) Assign the configuration (R or S) of each chiral center in coibacin B.
(b) Identify the number of possible stereoisomers for this compound, assuming that the geometry of the
alkenes is fixed.
Chapter 5 Solutions
Organic Chemistry (9th Edition)
Ch. 5.2 - Determine whether the following objects are chiral...Ch. 5.2A - Prob. 5.2PCh. 5.2B - Prob. 5.3PCh. 5.2B - Prob. 5.4PCh. 5.2C - Prob. 5.5PCh. 5.3 - Prob. 5.6PCh. 5.3 - Prob. 5.7PCh. 5.4D - Prob. 5.8PCh. 5.4D - Prob. 5.9PCh. 5.4D - Prob. 5.10P
Ch. 5.5 - Prob. 5.11PCh. 5.7 - When optically pure (R)-2-bromobutane is heated...Ch. 5.7 - Prob. 5.13PCh. 5.8 - Prob. 5.14PCh. 5.9B - Draw three-dimensional representations of the...Ch. 5.10A - For each sot of examples, make a model of the...Ch. 5.10A - Draw a Fischer projection for each compound....Ch. 5.10B - Prob. 5.18PCh. 5.10C - For each Fischer projection, label each asymmetric...Ch. 5.11C - Prob. 5.20PCh. 5.13 - Prob. 5.21PCh. 5.13 - Prob. 5.22PCh. 5.15 - Prob. 5.23PCh. 5.16A - Prob. 5.24PCh. 5 - The following four structures are naturally...Ch. 5 - For each structure, 1. star () any asymmetric...Ch. 5 - Prob. 5.27SPCh. 5 - Prob. 5.28SPCh. 5 - Prob. 5.29SPCh. 5 - Prob. 5.30SPCh. 5 - Prob. 5.31SPCh. 5 - Prob. 5.32SPCh. 5 - Prob. 5.33SPCh. 5 - Prob. 5.34SPCh. 5 - For each structure, 1. draw all the stereoisomers....Ch. 5 - Prob. 5.36SPCh. 5 - Prob. 5.37SPCh. 5 - 3,4-Dimethylpent-1-ene has the formula...Ch. 5 - A graduate student was studying enzymatic...Ch. 5 - Prob. 5.40SPCh. 5 - Prob. 5.41SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Draw a Lewis structure for each covalent molecule. a. HBr b. CH3F c. H2O2 d. N2H4 e. C2H6 f. CH2Cl2
Principles of General, Organic, Biological Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the absolute configuration of the following compounds anc draw the structure (Fischer projection) of the other enantiomer. F CH3 CN HO I`CH2NH2 H Br "CH3 CI H A В SH OH H3C" CH3 ČH2OH -C三CH ČH3 E HOOC Darrow_forwardNH2 N CH2 CH2 The stereochemistry around this chiral carbon is: O cannot be determined. O Rand S OR OSarrow_forward10- Two structures of glucose are shown below. CH₂OH HO HO OH LHa OH a-D-glucose HO,,. HO f) Draw the mirror image of a-D-glucose. d) How many possible stereoisomers? e) What is the relationship between a-D-glucose and B-D-glucose? CH₂OH OH OH Ha B-D-glucose Enantiomers/ diastereomers / conformers / constitutional / isomersarrow_forward
- a) Which of the following monosaccharides will react with Tollens' reagent? Circle all that аpply. СООН CH2OH CH2OH OCH3 но- H- ОН HO FH OH CH2OH CH2OH OH II III IV V b) Decide whether the disaccharide shown below is a reducing or non-reducing sugar CH2OH но Но- ОН CH2 но Но OH OH 우 오arrow_forwardDraw the stereoisomers of the following molecules in flying wedge representation and Fischer projection. HO OH H2N HO A Вarrow_forwardThe image shows Fischer projections for the structures of four isomers of a ketopentose. CH₂OH CH₂OH CH₂OH ПА пс C=O D HOEC H HOECH CH,OH A C=O H➡ COH но-с-н CH₂OH Which of the structures are stereoisomers of B? B Which of the structures are diastereomers of D? C=O HOI-C-H HOI-C-H CH₂OH с 000 ΤΑ B HO D CH₂OH O Which of the structures are enantiomers of C? H HI COH D CH₂OHarrow_forward
- 1. Below is the Fischer projection of a molecule. Is this the (R) or (S) enantiomer? 2. If you swap the positions of the H and the Cl, does this represent the same enantiomer or the opposite one? 3. Having swapped the positions of the H and CI, if you then swap the Cl and methyl group, what happens to the stereochemistry of the chiral center? CH3 H- CI CH,CH3arrow_forwardPredict the expected major product(s) of the following reaction sequence. ی ہیں میں قبر ہیں H 1. t-BuOK, t-BuOH 2. O3, CH2Cl2, -78 °C 3. MezS H OH OH + enantiomer Harrow_forwardWhat kind of isomers are the compounds shown in the box? A) Diastereomers B) Conformers C) Constitutional Isomers D) Enantiomers ОА овarrow_forward
- 1. How are each of the structures A-F related to structure X? They could be E (enantiomers), D C (conformers), or I (identical). (diastereomers), CH3 CI -Br HOH₂C- -OH CI X НО, H3C H₂C D CI Br CH₂OH CI HO STEREOISOMERS Br A CH2OH CI Br CI HOH₂COH CI CI Cl CH3 OH E -CH3 -CH₂OH B Br Br НО. Br CH3 HOH₂C CI C CI CH3 HO CH₂OH Farrow_forwardStudy Guide Questions Q7. Identify each Fischer projection as the D- or L-isomer H H - OH H-OH H-OH H-OH CH2OH HO- H HO CH₂OH C=0 - H OH -H CH₂OHarrow_forwardClassify the structures as being either an enantiomer, diastereomer or diastereomer/epimer of D-glucose. Structure A: CH H- -OH Structure B: но- H- H- OH Structure C: H- он H- -H D-Glucose: OH || CH CH CH он H- он но -H H- он но он но но- H- но H- OH OH но -- H- -H H -Ç-H Structure A: OH Structure B: OH Structure C: OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License