
Name each compound depicted in the ball-and-stick models.
a. 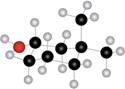 b.
b. 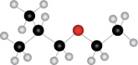 c.
c. 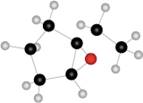
(a)
Interpretation: The name of given compound depicted in the ball-and-stick model is to be stated.
Concept introduction: One should follow the given steps to give the IUPAC name of cyclic alcohol. The first step is naming of ring that contains the carbon bonded to the hydroxyl group. While naming, the -e ending of parent cycloalkane must changed to the suffix -ol. The second step is numbering of carbon chain by providing lowest number to the
Answer to Problem 36P
The name of the given compound depicted in the ball-and-stick model is
Explanation of Solution
The given structure of alcohol is in the form of ball-and-stick model. It is converted into skeletal structure by replacing black ball with
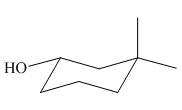
Figure 1
One should follow the given steps to give the IUPAC name of cyclic alcohol. The first step is naming of ring that contains the carbon bonded to the hydroxyl group. While naming, the -e ending of parent cycloalkane must changed to the suffix -ol. The second step is numbering of carbon chain by providing lowest number to the
The above structure of cyclic alcohol shows that it consists of
Thus, the name of the given compound depicted in the ball-and-stick model is
The name of the given compound depicted in the ball-and-stick model is
(b)
Interpretation: The name of given compound depicted in the ball-and-stick model is to be stated.
Concept introduction: One should follow the given steps to give the IUPAC name of ether. The first step is naming of longest chain as an alkane and the shorter chain as an alkoxy group. The second step is applying the other nomenclature rules.
Answer to Problem 36P
The name of the given compound depicted in the ball-and-stick model is
Explanation of Solution
The given structure of ether is in the form of ball-and-stick model. It is converted into skeletal structure by replacing black ball with
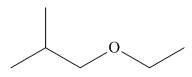
Figure 2
One should follow the given steps to give the IUPAC name of ether. The first step is naming of longest chain as an alkane and the shorter chain as an alkoxy group. The second step is applying the other nomenclature rules.
The above structure of ether shows that it consists of
Thus, the name of the given compound depicted in the ball-and-stick model is
The name of the given compound depicted in the ball-and-stick model is
(c)
Interpretation: The name of given compound depicted in the ball-and-stick model is to be stated.
Concept introduction: One should follow the given steps to give the IUPAC name of an epoxyalkane. The first step is naming of alkane chain or ring to which oxygen atom is bonded, and using the epoxy (prefix) to name epoxide. The second step is designating the location of the atoms (using two numbers) to which the oxygen atom is bonded.
Answer to Problem 36P
The name of the given compound depicted in the ball-and-stick model is
Explanation of Solution
The given structure of epoxide is in the form of ball-and-stick model. It is converted into skeletal structure by replacing black ball with
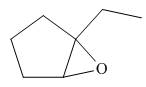
Figure 3
One should follow the given steps to give the IUPAC name of an epoxyalkane. The first step is naming of alkane chain or ring to which oxygen atom is bonded, and using the epoxy (prefix) to name epoxide. The second step is designating the location of the atoms (using two numbers) to which the oxygen atom is bonded.
The above structure of epoxide shows that the cyclopentane ring is bonded to the epoxide ring on
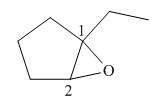
Figure 4
Thus, the name of the given compound depicted in the ball-and-stick model is
The name of the given compound depicted in the ball-and-stick model is
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry (6th Edition)
Additional Science Textbook Solutions
Chemistry: Atoms First
Chemistry: Structure and Properties (2nd Edition)
Introduction to Chemistry
Chemistry: Structure and Properties
Introductory Chemistry (6th Edition)
Chemistry: A Molecular Approach (4th Edition)
- Mestranol, sold under the brand names Enovid, Norinyl, and Ortho-Novum among others, is an estrogen medication which has been used in birth control pills. Select all of the functional group families, to which mestranol belongs. Choose one or more: A. Ester B. Alkene C. Ether D. Thiol E. Alcohol F. Arene G.Aldehyde H.Ketone I. Alkynearrow_forward(b). B. For each of the following, draw a líne structure of a molecule that contains the given functional groups and is consistent with the given molecular formula. Calculate the HDI (hydrogen deficiency index) and label the functional groups.arrow_forwardGlycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forward
- Name each compound. A. B. C. CH₂CH3 CH₂CH3 a & CI CI CI Br. ď OH CI compound A name: compound B name: compound C name:arrow_forwardexplain the following observations a. propanol is more water soluble than propane b. propanol is more water soluble than propanonearrow_forwardEncircle the functional groups, label each functional group with a letter, and identify the type or class of compounds represented by each functional group. 1. LSD, a hallucinogenic drug widely believed to be the inspiration behind the Beatles hit “Lucy in the Sky with Diamonds” 2. Methyljasmonate, a compound that belongs to a group called the pheromones. The male oriental fruit moth, Grapholitha molesta Busk., responds when a female moth emits this compound. 3.arrow_forward
- 1. Write the IUPAC name of each compound. a. Br b. C.arrow_forwardName attached compound depicted in the ball-and-stick models.arrow_forwardHow can phenol be distinguished from cyclohexanol? O A. solubility in water B. solubility in hydrochloric acid solution C. solubility in sodium bicarbonate solution D. solubility in sodium hydroxide solutionarrow_forward
- 1. Provide the IUPAC name. a. b. H3CO. H₂N D O₂N F NO2 IUPAC name IUPAC namearrow_forwardA sample consists of butyric acid, acetic acid and ethylene glycol. At a distilling temperature of 162°C, the distillate will significantly consist of: I. Butyric acid (boiling point: 162°C)II. Acetic acid (boiling point: 118°C)III. Ethylene glycol (boiling point: 197°C)A. I and II only B. I onlyC. I,II, and IIID. III onlyarrow_forward1. List the reasons why pentanol has a higher boiling point when compared to its corresponding alkane, pentane.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

