
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.7, Problem 9.17YT
Skill Building Kevlar®
Kevlar® is a polyamide used in bulletproof vests and the cases of some cell phones. Like PET, one of the monomers is terephthalic acid. The other monomer, phenylenediamine, contains two
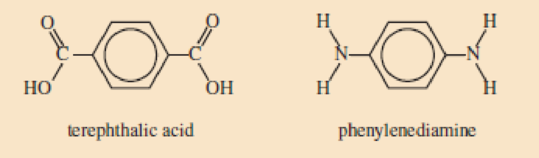
Draw a segment of a Kevlar® molecule built from two of each of these monomers.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Exercise 7) Make a molecular model of C2H6O. Now make another molecular model that is
different from the first one (but don't take that one apart yet).
Draw the structural formulas for both of your models in the space below, then write the
condensed formula below each structure.
Exercise 12) Draw the structural formulas for all of the structural isomers of C4H8.
Hint: There are six structural isomers. Using models can be very helpful with this one. Remember,
to be an isomer you have to actually break apart a bond. If you can simply rotate one of your
models and get the same shape it is not an isomer (that's called a conformation)!
Hint 2: Not every structure has a double bond!
Exercise:
Write formulas of each of the following compounds.
1.
Krypton trioxide
Iodine pentachloride
4.
2.
Carbon disulfide
Nitrogen trifluoride
5.
Diphosphorus pentoxide
Oxygen dibromide
6.
3.
Chapter 9 Solutions
Chemistry In Context
Ch. 9.1 - Scientific Practices Tennis Anyone? Examine this...Ch. 9.3 - Prob. 9.2YTCh. 9.3 - Prob. 9.3YTCh. 9.4 - Prob. 9.4YTCh. 9.4 - Prob. 9.5YTCh. 9.4 - Prob. 9.6YTCh. 9.4 - Prob. 9.7YTCh. 9.4 - Prob. 9.8YTCh. 9.4 - Prob. 9.9YTCh. 9.5 - Prob. 9.10YT
Ch. 9.5 - Skill Building Benzene and Phenyl The difference...Ch. 9.5 - Prob. 9.13YTCh. 9.5 - Skill Building Polystyrene Possibilities Show the...Ch. 9.6 - Skill Building Esters and Polyesters You have seen...Ch. 9.6 - Prob. 9.16YTCh. 9.7 - Skill Building Kevlar Kevlar is a polyamide used...Ch. 9.8 - Prob. 9.20YTCh. 9.8 - Your Turn 9.22 Skill Building Burning a Plastic...Ch. 9.8 - Your Turn 9.23 Scientific Practices Landfill...Ch. 9.9 - Examine the values in Table 9.4 from the American...Ch. 9.9 - Prob. 9.25YTCh. 9.9 - Prob. 9.26YTCh. 9.9 - Prob. 9.28YTCh. 9.10 - Skill Building The Chemistry of PLA We dont show...Ch. 9.11 - Your Turn 9.31 Scientific Practices Glass or...Ch. 9.11 - Prob. 9.32YTCh. 9.11 - Skill Building Meet DEHP DEHP belongs to a common...Ch. 9 - Prob. 1QCh. 9 - Prob. 2QCh. 9 - Prob. 3QCh. 9 - Prob. 4QCh. 9 - Prob. 5QCh. 9 - Prob. 6QCh. 9 - Prob. 7QCh. 9 - Prob. 8QCh. 9 - Prob. 9QCh. 9 - Prob. 10QCh. 9 - Prob. 11QCh. 9 - Prob. 12QCh. 9 - Prob. 13QCh. 9 - Prob. 14QCh. 9 - Prob. 15QCh. 9 - Prob. 16QCh. 9 - Prob. 17QCh. 9 - Prob. 18QCh. 9 - Prob. 19QCh. 9 - Prob. 20QCh. 9 - Prob. 21QCh. 9 - Prob. 22QCh. 9 - Prob. 23QCh. 9 - Prob. 24QCh. 9 - Prob. 25QCh. 9 - Prob. 26QCh. 9 - Prob. 27QCh. 9 - Prob. 28QCh. 9 - Prob. 29QCh. 9 - Prob. 30QCh. 9 - Prob. 31QCh. 9 - Prob. 32QCh. 9 - Prob. 33QCh. 9 - Prob. 34QCh. 9 - Prob. 35QCh. 9 - Prob. 36QCh. 9 - Prob. 37QCh. 9 - Prob. 38QCh. 9 - Prob. 39QCh. 9 - Prob. 40QCh. 9 - Prob. 41QCh. 9 - Prob. 42QCh. 9 - Prob. 43QCh. 9 - Prob. 44QCh. 9 - Prob. 45QCh. 9 - Prob. 46QCh. 9 - Prob. 47QCh. 9 - Prob. 48QCh. 9 - Prob. 49QCh. 9 - Prob. 50QCh. 9 - Prob. 51QCh. 9 - Prob. 52QCh. 9 - Prob. 53QCh. 9 - Prob. 54QCh. 9 - Prob. 55QCh. 9 - Prob. 56QCh. 9 - Prob. 57QCh. 9 - Prob. 58QCh. 9 - Prob. 59Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Two molecules, each with the general formula AX3, have different dipole moments. Molecule Y has a dipole moment of zero, whereas molecule Z has a nonzero dipole moment. From this information, what can you say about the geometries of Y and Z?arrow_forwardWashing soda has the formula Na2CO310H2O. What is the chemical name of this substance?arrow_forwardExercise 12) Draw the structural formulas for all of the structural isomers of C4Hg. Hint: There are six structural isomers. Using models can be very helpful with this one. Remember, to be an isomer you have to actually break apart a boıd. If you can simply rotate one of your models and get the same shape it is not an isomer (that's called a conformation)! Hint 2: Not every structure has a double bond!arrow_forward
- Exercise 2: Carbon Containing Oxygen Molecules 1. Why is ethanol (CH₂CH₂OH) more soluble in water than 1- (CH₂CH₂CH₂CH₂CH₂CH₂OH)? hexanol 2. Why does 1-butanol (CH CH₂CH₂CH₂OH) have a lower boiling point than 1-hexanol (CH₂CH₂CH₂CH₂CH₂CH₂OH)?arrow_forwardPractice question helparrow_forwardОН ОН What is this molecule's chemical formula? Note: write the simplest molecular chemical formula, in which each element symbol appears only once. CH, |CH, How many CH,, CH,, and CH groups are in this molecule? CH Explanation Check O2022 McGraw Hill LLC. All Ric DELLarrow_forward
- Dinitrogen difluoride (see Example 10.5) exists as cis and trans isomers. Write structural formulas for these isomers and explain (in terms of the valence bond theory of the double bond) why they exist.arrow_forwardExercise 1 Look at the MSDS and containers for four common laboratory chemicals: ethanol (denatured laboratory alcohol), sodium chloride, sodium hydroxide, and formaldehyde. List the NFPA hazards for each. Chemical Name Hazards ethanol sodium chloride sodium hydroxide formaldehydearrow_forwardEstimate the OH bond length in H2O from the covalent radii listed in Table 9.4.arrow_forward
- Exercise 9) Draw the structural formulas for all of the possible structural isomers of C4H10. Write the condensed formula below each structure. Hint: There are two structural isomers. You may find it helpful to build models to solve this exercise.arrow_forwardAnswer the questions in the table below about this molecule: ? What is this molecule's chemical formula? Note: write the simplest molecular chemical formula, in which each element symbol appears only once. CH; CH, How many CH,, CH,, and CH groups are in this molecule? CH Check Explanation 2022 McGraw Hill LLC, All Rights Reserved. Terms of Use | Privacy Cer W DELL ESC F1 F2 F3 F4 F5 F6 F7 F8 Fnarrow_forwardAnswer the questions in the table below about this molecule: Но OH What is this molecule's chemical formula? Note: write the simplest molecular chemical formula, in which each element symbol appears only once. CH, How many CH,, CH,, and CH groups are in this molecule? CH, CH Explanation Check O 2022 McGraw Hill LLC. All Rights Reserved. Terms of W DELLarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY