Q: A piece of an unknown metal with a mass of 23.8g is heated to 100.0°C and dropped in 50.0g of water…
A: The specific heat of a substance is the amount of heat needed to raise the temperature of of the…
Q: OMe OMe OH HO HO- HO OMe H+ ong HO- HO OMe Pre-practical "MUST DO" to be included in your lab-book:…
A: The objective of the question is to understand the pre-practical steps to be included in the…
Q: A. image click here Perform the retrosynthetic analysis for the indicated bond and choose the…
A: This is an example of nucleophilic addition of alkyne to aldehyde
Q: explain how mass spectrometer operates describing how particles are selected
A: This question aims to explain the work of a mass spectrometer and describe how the particles are…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A:
Q: Draw a structural formula for the major organic product of the following reaction:
A: When an alkene reacts with Br2 in the presence of CH2Cl2, a dibromo alkane will be formed.This…
Q: Он + NaOH
A: This reaction is an example of acid-base reaction.
Q: Draw the structures of the products, showing stereochemistry where relevant. 3. a)…
A: These are the examples of electrophilic addition reactions.
Q: please show the correct arrows that will create the product Mg ^ Select to Edit Arrows H3O+ H' H H…
A: Information about the question
Q: Name: Give the IUPAC name for each compound. Part 1 of 2 Give detailed Solution with explanation…
A: IUPAC nomenclature are the set of rules that are used or taken into consideration while naming any…
Q: 7. Draw the product of the following reaction. H3C-N H3C CH3
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: With reference to the H-NMR spectra given below, answer the following: For the starting material,…
A:
Q: Question 3 Draw Zaitsev and Hofmann products that are expected when each of the following compounds…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Use the standard reduction potentials located in the 'Tables' linked above to calculate the…
A:
Q: Why do we use dilute HCl for the first step and concentrated HCl for the second step? о OH NaBH HCI…
A: Secondary alcohols can undergo SN2 and SN1 reactions to form alkyl halide:Dilute HCl (aqueous): In…
Q: Provide an acceptable name for the following compound. 0- CH-N-CH2CH3 Spell out the full name of the…
A: RULES for IUPAC nomenclature; In first step, we identify functional groups present. We make longest…
Q: An analytical chemist is titrating 109.4 mL of a 0.7700 M solution of hydrazoic acid (HN3) with a…
A: We will first calculate the millimoles of both acid and base. Then we will find whether weak acid or…
Q: Provide reagents and conditions for the following synthetic tranformation. (Please don't provide…
A: In the given reaction the alcohol is converted into carboxylic acid and an extra carbon atom is…
Q: Draw structural formulas for the a,ẞ-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: The product formed is given as,
Q: Which of the following terms best describes the pair of compounds shown: enantiomers, diastereomers,…
A:
Q: Calculate the percentage by mass of oxygen in the following compounds. 4 Part B methadone, C21H27 NO…
A:
Q: Show the starting diene and dienophile you could use to prepare the following molecule: Diene +…
A: Given,The reaction is:
Q: Consider the following reaction:COCl2(g) CO(g) + Cl2(g)If 2.98×10-2 moles of COCl2, 0.206 moles of…
A: Kp=Kc(RT)ΔnKc=[COCl2][CO][Cl2]=17.8L2.98x10−2moles17.8L(0.206moles)x17.8L(0.304moles)=0.118…
Q: The Kb of hydroxylamine, NH2OH, is 1.10×10−8. A buffer solution is prepared by mixing 100.0 mL of a…
A: pKb = - log Kb = - log (1.10 * 10-8 ) = 7.96pKa = 14-pKb = 14- 7.96…
Q: What is the major product of the following reaction? 1) B₂H6, diglyme 2) H₂O2, NaOH
A:
Q: Consider the following three compounds. Part: 0/2 Part 1 of 2 Rank the three compounds in order of…
A: We need to determine the order of heat of hydrogenation for given alkenes.Organic compounds that…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the alcohol side product.…
A: Acid catalyzed hydrolysis of ester results carboxylic acid and alcohol
Q: Under constant-volume conditions the heat of combustion of benzoic acid (CH3COOH) is -26.38 kJ/g. A…
A: Given,the heat of combustion of benzoic acid = - 26.38 kJ/g (- ve sign indicates that heat is…
Q: One cup of fresh orange juice contains 139 mg of ascorbic acid (vitamin C, C6H8O6). Part A Given…
A: Given:Mass of ascorbic acid in one cup of fresh orange juice = 139 mg = 0.139 g Volume of one cup =…
Q: 5. (2Z, 4S)-4-Bromohept-2-enal 6. P-Nitrobenzaldehyde
A:
Q: Draw a structural formula of the SR configuration of the compound shown below. HO CH3 -NHCH3 R H3C…
A:
Q: How many unpaired electrons are in a chlorine atom? (Please 1 step is introduction and all step…
A: Filling of electrons in atomic orbital follow Aufbau principle.It states that electrons are filled…
Q: The Ksp of Ca(OH)2 is 5.066 × 10–5 at 25 °C. What is the concentration of OH–(aq) in a saturated…
A: The reaction is:
Q: Identifying Highlight in red the primary carbons in this molecule. Highlight the tertiary carbons in…
A: Any carbon atom attached to one other carbon atom or no other carbon atom is called a primary carbon…
Q: Which statements are true about weak acids? They dissociate completely in water. The Ka values are…
A: The objective of the question is to identify the correct statements about weak acids among the given…
Q: Consider the following equilibrium system: 2CrO4 2-(aq)+2H+(aq) = Cr2O72 (aq)+H2O(1) In what…
A: As per Le-Chatelier's principle, when a stress is applied to a system at equilibrium, the…
Q: What is the expected major product for the following reaction? 1. Hg(OAc)2, H₂O 2. NaBH4, NaOH 01 ON…
A:
Q: If 36.7 g of AgNO₃ react with 28.6 g of H₂SO₄ according to this UNBALANCED equation below, how many…
A: The objective of this question is to determine the amount of Ag2SO4 that could be formed from the…
Q: 4. Draw a mechanism for the reaction below. CH3NH2
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 39. Which set of protons corresponds to the quintet at 4.5 ppm? Note: Equivalent protons are color…
A: spin-spin splittingnumber of peaks =n = number of neighboring hydrogen.In the NMR spectrum, we get…
Q: question 12 help
A: Answers given below Explanation:
Q: 4. Which alkyl halide would be most reactive in an SN1 reaction? Explain your answer. I Br IV Br Br…
A: The electrical effect is particularly important for the SN1 reaction. An SN1 reaction involves…
Q: What is the pH of a 0.950 M solution of Na2HPO4? H3PO4 is a weak polyprotic acid with Ka1 =…
A: The objective of this question is to calculate the pH of a 0.950 M solution of Na2HPO4. The compound…
Q: Calculate a value for the equilibrium constant for the reaction O2(g) + O (g) = O3(g) given…
A: The objective of the question is to find the equilibrium constant for the reaction O2(g) + O (g) =…
Q: Diethylamine (CH3CH2)2NH is used in many pharmaceuticals. The Ka of (CH3CH2)2NH2* is 1.2 × 10- at…
A: We will first calculate the value of Kb from Ka by using the formula -Then we will construct ICE…
Q: excess CH3OH H+
A: This is an example of ketal formation reaction
Q: K Problem 12 of 14 Submit Draw the missing organic structures or select the missing reagents in the…
A: Starting Materials:PhCHO (phenyl ethanol)H3O+ (hydronium ion)PBr3 (phosphorus tribromide)DMF…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. KCN 2. LiAlH4 (excess) 3.…
A: In this reaction we have to convert alkyl bromide into alkane.
Q: Predict the major product for the following reaction. مومو H (xs) ? Modify the given structure of…
A: Carboxylic acid derivatives such as amides, esters, acid halides and anhydrides generally undergo…
Q: Measured Mass of NaCI: 11.70 g Measured Volume of Water: 100.0 mL Answer each question with correct…
A: Information about the question
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

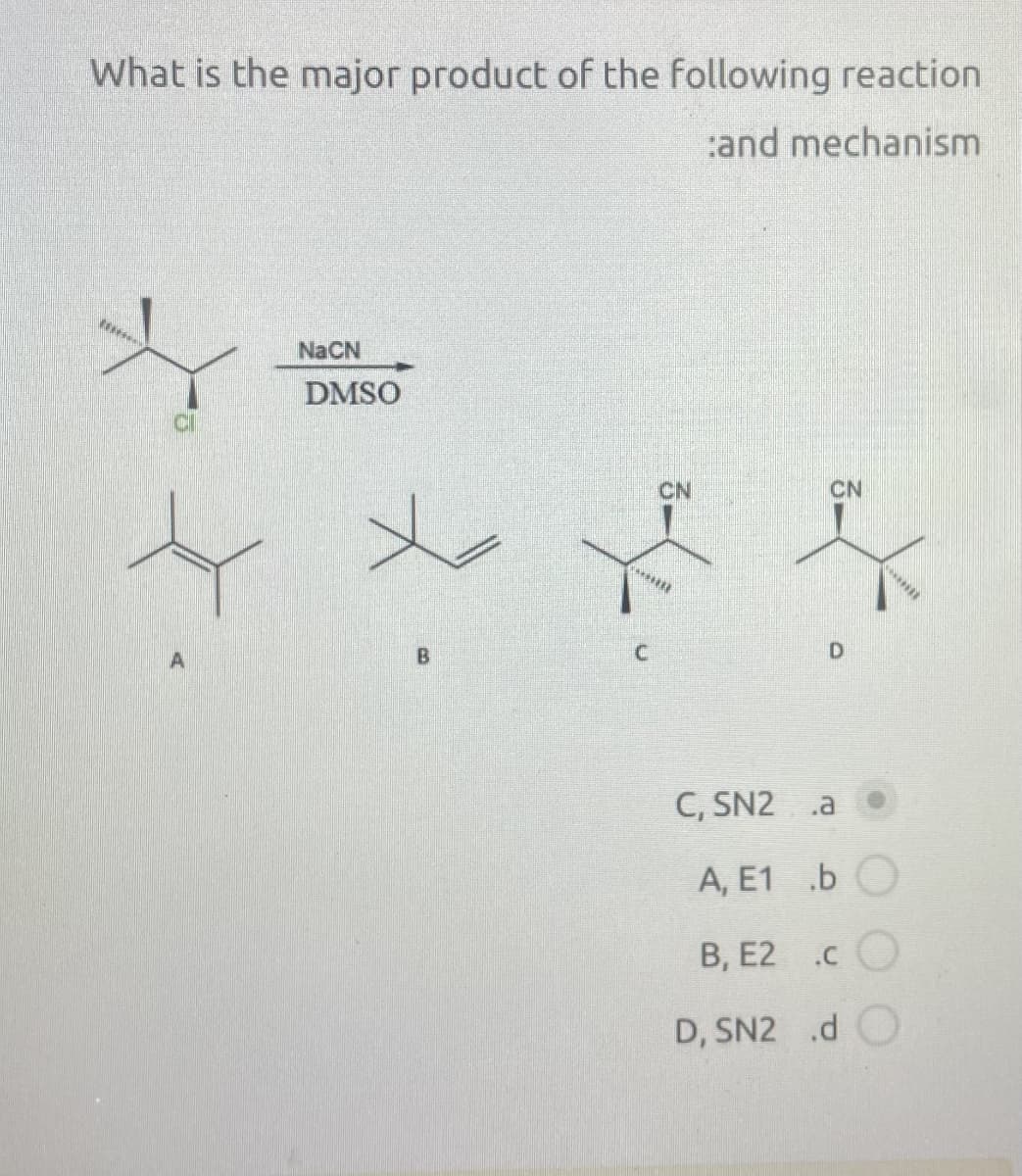
- Two chemists at Dupont found that ICH2ZnI is better than diazomethane at converting a C=C bond to a cylcopropane ring. Propose a mechanism for the reaction, now known as the Simmons–Smith reaction in their honor.Substitutions vs elimination. Pls help me identify the major products and the dominant mechanism. (SN1/SN2/E1/E2 or their combination)A carbocation intermediate is central to which of these reaction types? E1, E2, SN1, SN2 ? Choose 1
- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?Predict the product of the following reaction and classify the reaction. Pb(NO3)2+FeSO4--->PbSO4+________The following halide reacts differently with hydrogen sulfide as shownshown in the attached image reactions. For each reaction, show the mechanism, draw the energy diagrams for the formation of each product and write the rate equation for the formation of each product. PD: mechanisms are SN1,SN2,E1 etc
- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- ΔH=-75 kJ/mol Rate = k [CH3CH2Br][CH3COO-] Which reaction energy profile would be the best representative of the data provided?The replacement of CH3OH to dimethyl sulfoxide, DMSO (CH3)2S=O) as a solvent in the substitution reaction below results in what?Identify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2

