
University Physics Volume 2
18th Edition
ISBN: 9781938168161
Author: OpenStax
Publisher: OpenStax
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 1, Problem 30CQ
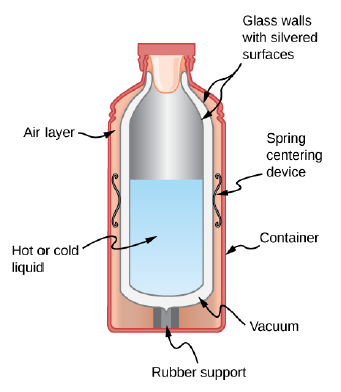
Shown below is a cut-away drawing of a thermos bottle (also known as a Dewar flask), which is a device designed specifically to slow down all forms of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The figure shows a cut-away drawing of a thermos bottle (also known as a Dewar flask), which is a device designed specifically to slow down all forms of heat transfer. Explain the functions of the various parts, such as the vacuum, the silvering of the walls, the thin-walled long glass neck, the rubber support, the air layer, and the stopper.
You have a Dewar flask (a laboratory vacuum flask) that has an open top and straight sides, as shown below. You fill it with water and put it into the freezer. It is effectively a perfect insulator, blocking all heat transfer, except on the top. After a time, ice forms on the surface of the water. The liquid water and the bottom surface of the ice, in contact with the liquid water, are at . The top surface of the ice is at the same temperature as the air in the freezer, Set the rate of heat flow through the ice equal to the rate of loss of heat of fusion as the water freezes. When the ice layer is 0.700 cm thick, find the rate in m/s at which the ice is thickening.
A 6.0-cm-diameter cylinder of nitrogen gas has a 4.0-cm-thick movable copper piston. The
cylinder is oriented vertically, as shown in the figure, and the air above the piston is evacuated.
When the gas temperature is 25 °C, the piston floats 20 cm above the bottom of the cylinder.
(Figure 1)
Figure
20 cm
Vacuum
Piston.
Ź
6 cm
1 of 1
Then 1.5 J of heat energy are transferred to the gas. What is the new equilibrium temperature of the gas in °C?
Express your answer in degrees Celsius.
► View Available Hint(s)
T₂ = 115.06992
Submit
Part D
[Π| ΑΣΦ
X Incorrect; Try Again; 5 attempts remaining
Submit
What is the final height of the piston?
Express your answer with the appropriate units.
► View Available Hint(s)
Previous Answers
LO
L₂= 28.05
Part E
6
HÅ
Previous Answers
cm
μÀ
?
X Incorrect; Try Again; 5 attempts remaining
How much work is done on the gas as the piston rises?
Express your answer with the appropriate units.
► View Available Hint(s)
?
?
°C
Chapter 1 Solutions
University Physics Volume 2
Ch. 1 - Check Your Understanding Does a given reading on a...Ch. 1 - Check Your Understanding Two objects A and B have...Ch. 1 - Check Your Understanding If 25 kJ is necessary to...Ch. 1 - Check Your Understanding Explain why a cup of...Ch. 1 - Check Your Understanding Why does snow Often...Ch. 1 - Check Your Understanding Name an example from...Ch. 1 - Check Your Understanding How does the rate of heat...Ch. 1 - Check Your Understanding Explain why using a fan...Ch. 1 - Check Your Understanding How much greater is the...Ch. 1 - What does it mean to say that two systems are in...
Ch. 1 - Give an example in which A has some kind of...Ch. 1 - If a thermometer is allowed to come to equilibrium...Ch. 1 - Give an example of a physical property that varies...Ch. 1 - Pouring cold water into hot glass or ceramic...Ch. 1 - One method of getting a tight fit, say of a metal...Ch. 1 - Does it really help to run hot water over a tight...Ch. 1 - When a cold alcohol thermometer is placed in a hot...Ch. 1 - Calculate the length of a 1-meter rod of a...Ch. 1 - Noting the large stresses that can be caused by...Ch. 1 - How is heat transfer related to temperature?Ch. 1 - Describe a situation in which heat transfer...Ch. 1 - When heat transfers into a system, is the energy...Ch. 1 - The brakes in a car increase in temperature by T...Ch. 1 - A pressure cooker contains water and steam in...Ch. 1 - As shown below, which is the phase diagram for...Ch. 1 - Can carbon dioxide be liquefied at room...Ch. 1 - What is the distinction between gas and vapor?Ch. 1 - Heat transfer can cause temperature and phase...Ch. 1 - How does the latent heat of fusion of water help...Ch. 1 - What is the temperature of ice right after it is...Ch. 1 - If you place 0 ice into 0 water in an insulated...Ch. 1 - What effect does condensation on a glass of ice...Ch. 1 - In Miami, Florida, which has a very humid climate...Ch. 1 - In winter, it is often warmer in San Francisco...Ch. 1 - Freeze-dried foods have been dehydrated in a...Ch. 1 - In a physics classroom demonstration, an...Ch. 1 - Mechanisms of Heat Transfer What are the main...Ch. 1 - When our bodies get too warm, they respond by...Ch. 1 - Shown below is a cut-away drawing of a thermos...Ch. 1 - Some electric stoves have a flat ceramic surface...Ch. 1 - Loose-fitting white clothing covering most of the...Ch. 1 - One way to make a fireplace more energy-efficient...Ch. 1 - On cold, clear nights horses will sleep under the...Ch. 1 - When watching a circus during the day in a large,...Ch. 1 - Satellites designed to observe the radiation from...Ch. 1 - Why are thermometers that ale used in weather...Ch. 1 - Putting a lid on a boiling pot greatly reduces the...Ch. 1 - Your house will be empty for a while in cold...Ch. 1 - You pour coffee into an unlidded cup, intending to...Ch. 1 - Broiling is a method of cooking by radiation,...Ch. 1 - On a cold winter morning, why does the metal of a...Ch. 1 - While traveling outside the United States, you...Ch. 1 - What are the following temperatures on the Kelvin...Ch. 1 - (a) Suppose a cold front blows into your locale...Ch. 1 - An Associated Press article on climate change...Ch. 1 - (a) At what temperature do the Fahrenheit and...Ch. 1 - A person taking a reading of the temperature in a...Ch. 1 - The height of the Washington Monument is measured...Ch. 1 - How much taller does the Eiffel Tower become at...Ch. 1 - What is the change in length of a 3.00-cm-long...Ch. 1 - How large an expansion gap should be left between...Ch. 1 - You are looking to buy a small piece of land in...Ch. 1 - Global warming will produce rising sea levels...Ch. 1 - (a) Suppose a meter stick made of steel and one...Ch. 1 - (a) If a 500-mL glass beaker is filled to the brim...Ch. 1 - Most cars have a coolant reservoir to catch...Ch. 1 - A physicist makes a cup of instant coffee and...Ch. 1 - The density of water at 0 C is very nearly 1000...Ch. 1 - Show that =3a , by calculating the infinitesimal...Ch. 1 - Calorimetry On a hot day, the temperature of an...Ch. 1 - To sterilize a 50.0-g glass baby bottle, we must...Ch. 1 - The same heat transfer into identical masses of...Ch. 1 - Rubbing your hands together warms them by...Ch. 1 - A 0.250-kg block of a pule material is heated from...Ch. 1 - Suppose identical amounts of heat transfer into...Ch. 1 - (a) The number of kilocalories in food is...Ch. 1 - Following vigorous exercise, the body...Ch. 1 - In a study of healthy young men[1], doing 20...Ch. 1 - A 1.28-kg sample of water at 10.0 is in a...Ch. 1 - Repeat the preceding problem, assuming the water...Ch. 1 - How much heat transfer (in kilocalories) is...Ch. 1 - A bag containing 0 ice is much more effective in...Ch. 1 - (a) How much heat transfer is required to raise...Ch. 1 - Condensation on a glass of ice water causes the...Ch. 1 - On a trip, you notice that a 3.50-kg bag of ice...Ch. 1 - On a certain dry sunny day, a swimming pool 's...Ch. 1 - (a) How much heat transfer is necessary to raise...Ch. 1 - In 1986, an enormous iceberg broke away from the...Ch. 1 - How many grams of coffee must evaporate from 350 g...Ch. 1 - (a) It is difficult to extinguish a fire on a...Ch. 1 - The energy released from condensation in...Ch. 1 - To help prevent frost damage, 4.00 kg of water at...Ch. 1 - A 0.250-kg aluminum bowl holding 0.800 kg of soup...Ch. 1 - A 0.0500-kg ice cube at 30.0 is placed in 0.400...Ch. 1 - If you pour 0.0100 kg of 20.0 water onto a...Ch. 1 - Indigenous people sometimes cook in watertight...Ch. 1 - What would the final temperature of the pan and...Ch. 1 - (a) Calculate the rate of heat conduction through...Ch. 1 - The rate of heat conduction out of a window on a...Ch. 1 - Calculate the rate of heat conduction out of the...Ch. 1 - Suppose you stand with one foot on ceramic...Ch. 1 - A man consumes 3000 kcal of food in one day,...Ch. 1 - A firewalker runs across a bed of hot coals...Ch. 1 - (a) What is the rate of heat conduction through...Ch. 1 - A warms transfers energy by conduction through its...Ch. 1 - Compare the rate of heat conduction through a...Ch. 1 - Suppose a person is covered head to foot by wool...Ch. 1 - Some stove tops are smooth ceramic for easy...Ch. 1 - One easy way to reduce heating (and cooling) costs...Ch. 1 - Many decisions are made on the basis of the...Ch. 1 - In 1701, the Danish astronomer Ole Rømer proposed...Ch. 1 - What is the percent error of thinking the melting...Ch. 1 - An engineer wants to design a structure in which...Ch. 1 - How much stress is cleated in a steel beam if its...Ch. 1 - A brass rod (Y=90109N/m2), with a diameter of...Ch. 1 - A mercury thermometer still in use for meteorology...Ch. 1 - Even when shut down after a period of normal use,...Ch. 1 - You leave a pastry in the refrigerator on a plate...Ch. 1 - Two solid spheres, A and B, made of the same...Ch. 1 - In some countries, liquid nitrogen is used on...Ch. 1 - Some gun fanciers make their own bullets, which...Ch. 1 - A 0.800-kg iron cylinder at a temperature of...Ch. 1 - Repeat the preceding problem with 2.00 kg of ice...Ch. 1 - Repeat the preceding problem with 0.500 kg of ice,...Ch. 1 - A 30.0-g ice cube at its melting point is dropped...Ch. 1 - (a) Calculate the rate of heat conduction through...Ch. 1 - (a) An exterior wall of a house is 3 m tall and 10...Ch. 1 - For the human body, what is the rate of heat...Ch. 1 - You have a Dewar flask (a laboratory vacuum flask)...Ch. 1 - An infrared heater for a sauna has a surface area...Ch. 1 - (a) Deter-nine the power of radiation from the Sun...Ch. 1 - A pendulum is made of a rod of length L and...Ch. 1 - At temperatures of a few hundred kelvins the...Ch. 1 - In a calorimeter of negligible heat capacity, 200...Ch. 1 - An astronaut performing an extra-vehicular...Ch. 1 - The goal in this problem is to find the growth of...Ch. 1 - As the very first rudiment of climatology,...Ch. 1 - Let's stop ignoring the greenhouse effect and...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Choose the best answer to each of the following. Explain your reasoning. What happens if a white dwarf reaches ...
Cosmic Perspective Fundamentals
Write each number in scientific notation.
14. 1,400,000
Applied Physics (11th Edition)
Given Earths mass, the Moons distance and orbital period, and the value of G, could you calculate the Moons mas...
Essential University Physics (3rd Edition)
30. A 3000-rn-high mountain is located on the equator. How much faster does a climber on top of the mountain mo...
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Agent Bond is standing on a bridge, 13m above the road below, and his pursuers are gelling loo close for comfor...
Physics for Scientists and Engineers with Modern Physics
l. Suppose you have the uniformly charged cube in FIGURE Q24.1. Can you use symmetry alone to deduce the shape ...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- (a) Calculate the rate of heat conduction through a double-paned window that has a 150-m2 area and is made of two panes of 0.800 cm-thick glass separated by a 1.00 cm air gap. The inside surface temperature is 15.0 C, while that on the outside is 10.0 OC. (Hint: There are identical temperature drops across the two glass panes. First find these and then the temperature drop across the air gap. This problem ignores the increased heat transfer in the air gap due to convection.) (b) Calculate the rate of heat conduction through a 1.60-cm-thick window of the same area and with the same temperatures. Compare your answer with that for part (a).arrow_forwardA cut-away drawing of a thermos bottle (also known as a Dewar flask), which is a device designed specifically to slow down all forms of heat transfer. Explain the functions of the various parts, such as the vacuum, the silvering of the walls, the thin-walled long glass neck, the rubber support, the air layer, and the stopper.arrow_forwardTwo finned surfaces are identical, except that the convection heat transfer coefficient of one of them is twice that of the other. For which finned surface is the (a) fin effectiveness and (b) fin efficiency higher? Explain.arrow_forward
- a 6.0 cm diameter cyliinder of nitrogen gas has a 4.0 cm thick movable copper piston. the cylinder is oriented vertically as shown in the figure, and the air above the piston is evacuated. when the gas temperature is 25 degree C the piston floats 20 cm above the bottom of the cylinder then 1.5 J of heat energy are transferred to the gas. what is the new equilibrium temperature of the gas in degree Carrow_forwardWhen is the local heat transfer coefficient infinite? Why? When a flat plate is subjected to uniform heat flux, how different are the relations compared to the isothermal plate case? Questions above are related to Heat transferarrow_forwardYou have a Dewar flask (a laboratory vacuum flask) that has an open top and straight sides, as shown below. You fill it with water and put it into the freezer. It is effectively a perfect insulator, blocking all heat transfer, except on the top. After a time, ice forms on the surface of the water. The liquid water and the bottom surface of the ice, in contact with the liquid water, are at 0 °C . The top surface of the ice is at the same temperature as the air in the freezer, −18 °C. Set the rate of heat flow through the ice equal to the rate of loss of heat of fusion as the water freezes. When the ice layer is 0.700 cm thick, find the rate in m/s at which the ice is thickening.arrow_forward
- WITH THE CONCEPT OF HEAT TRANSFER, ANSWER THE FOLLOWING AND SHOW THE COMPLETE SOLUTION: A 10.0-cm long steel rod is welded end to end to a 20.0-cm long copper rod of the same cross-sectional area. The thermal conductivities of steel and copper are 50.2 W/m·K and 385 W/m·K, respectively. The steel end is kept at 100°C while the copper end is kept at 0°C. What is the steady-state temperature at the interface connecting the two rods?arrow_forwardA 58-kg cross-country skier glides over snow as in the figure below. The coefficient of friction between skis and snow is 0.24. Assume all the snow beneath her skis is at 0°C and that all the internal energy generated by friction is added to snow, which sticks to her skis until it melts. How far would she have to ski to melt 1.5 kg of snow? Your incorrect answer may have resulted from roundoff error. Make sure you keep extra significant figures in intermediate steps of your calculation. m Need Help? Read It Master itarrow_forwardDescribe the different mechanisms of heat transfer that affect the drying of washing outdoors. Is it better to have a sheltered sunny spot or a shaded windy location? For the toolbar, press ALT+F10 (PC) or ALT+FN+F10 (Mac). B IU S Paragraph Open Sans, s... v 10pt Ix •.. > !!! >arrow_forward
- A hollow cylinder has length L, inner radius R, and thickness d, and the temperatures at the inner and outer surfaces are Tn and Ti, respectively. The thermal conductivity of the material of which the cylinder is made is k. Derive an equation for the rate of heat transfer P through the walls of the cylinder. Simplify this equation assuming that the thickness of the cylinder d is much smaller than the its inner radius R.arrow_forwardThe 2010 Nobel Prize in Physics was awarded for the discovery of graphene, a two-dimensional form of carbon in which the atoms form a two- Part A dimensional crystal-lattice sheet only one atom thick. Predict the molar specific heat of graphene. Give your answer as a multiple of R. Express your answer in terms of R. ΑΣφ Carrow_forwardA insulating cylinder has an equal amount of gas on both sides of a movable piston, as shown in the above figure. Initially, the two compartments have equal volumes and their temperatures are 550 K and 250 K. The piston then moves slowly until it comes to rest at an equilibrium position. Find the final temperatures in the two portions.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning


Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY