
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
13th Edition
ISBN: 9780134421353
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 11.47APP
Give the IUPAC name for each of the following: (11.3)
a.
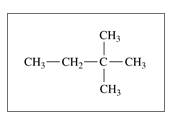
b. CH3 CH2
c.
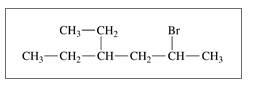
d.
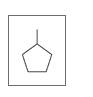
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
12.38 The compound frambinone has the taste of raspberries and
has been used in weight loss. Identify the functional groups in
frambinone.
(12.1, 12.3)
HO
O
CH,—CH,—C–CH3
14
Frambinone
odio
12.50 Draw the condensed or line-angle structural formula, if cyclic,
for each of the following compounds: (12.1, 12.2)
a. 2,3-dichlorocyclobutyl alcohol
C. 2-methyl-5-bromo-3-hexanol
b. butyl propyl ether
d. 2-methyl-4-nitrophenol
12.52 Draw the condensed structural or line-angle formula for the
alkene, aldehyde, or ketone product of each of the following
reactions: (12.4)
H, heat
a.
ОН
CH; OH
b. CHз — СH — СH-CH,
OH
[0]
ОН
d. CH3-CH2-CH2-CH-CH3
Chapter 11 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Prob. 11.5PPCh. 11.1 - Prob. 11.6PPCh. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Draw the condensed structural formula for alkanes...Ch. 11.2 - Draw the condensed structural formula for alkanes...
Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Give the IUPAC name for each of the following a....Ch. 11.3 - Give the TUPAC name for each of the following: a....Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the line-angle formula for each of the...Ch. 11.3 - Prob. 11.18PPCh. 11.4 - Heptane, used as a solvent for rubber cement, has...Ch. 11.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Draw the condensed structural formula, or...Ch. 11.5 - Prob. 11.28PPCh. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Draw the condensed structural formula for each of...Ch. 11.6 - Prob. 11.32PPCh. 11.7 - Draw the structural formula for the product in...Ch. 11.7 - Draw the structural formula for the product in...Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11 - Prob. 11.41UTCCh. 11 - Prob. 11.42UTCCh. 11 - Prob. 11.43UTCCh. 11 - Prob. 11.44UTCCh. 11 - Convert each of the following line-angle formulas...Ch. 11 - Convert each of the following line-angle formulas...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name (including cis or trans, if...Ch. 11 - Give the LUPAC name (including cis or trans, if...Ch. 11 - Prob. 11.51APPCh. 11 - Prob. 11.52APPCh. 11 - Name each of the following aromatic compounds:...Ch. 11 - Prob. 11.54APPCh. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Prob. 11.59APPCh. 11 - Draw the line-angle formula for each of the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Give the name for the product from the...Ch. 11 - Give the name for the product from the...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Prob. 11.67CPCh. 11 - Prob. 11.68CPCh. 11 - Prob. 11.69CPCh. 11 - Prob. 11.70CPCh. 11 - Prob. 11.71CPCh. 11 - Prob. 11.72CPCh. 11 - Prob. 11.73CPCh. 11 - Margarines are produced from the hydrogenation of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 12.42 Which of the following will give a positive Tollens' test? (12.4) 1-propanol b. 2-propanol c. hexanal a.arrow_forward12.60 Which of the following aldehydes or ketones are soluble in water? (12.3) |3| a. CH3-CH2 -C-CH3 b. CH3 — С —н CH3 c. CH3-CH2-CH-CH2-CH2-C-Harrow_forwardd. CH3-CH2-C-OH 16.58 Which of the following compounds are soluble in water? (16.2, 16.5) а. СH3 — СН, — СH, —С-ОН b. CH3-C-0-CH3 ers c. CH3-CH2-CH2- ОН acid Su d. .1, 16.59 Draw the condensed structural formulas for the products from each of the following reactions: (16.2, 16.3) daim a. CH3-CH2-C-OH + H,O A b. CH3-CH,-C-OH + KOH ОН c acid H, heat с. acid "ОН + С—ОН H*, heat + HO–CH2 CH3 2 d. 16.60 Draw the condensed structural formulas for the products from each of the following reactions: (16.2, 16.3) olone odw a. CH3-C -OH + NaOH || b. CHз— С— ОН + Н2О с. ОН + КОН O=arrow_forward
- (12.9) Which of the following has the highest boiling point? O Both H₂S and H₂O have the same boiling point that is higher than the boiling point of H₂Se. O H₂Se O H₂O O H₂Sarrow_forward10. Draw the structural formula for the product in each of the following addition reactions for an alkene: (11.7) a) CH3-CH2-C=CH-CH3 + H,O CH3-CH₂ b) CH 3 CH3-CH-C=CH-CH3 + H₂ CH3 + H₂ Pt H Niarrow_forward12:46 16.48 Give the IUPAC and common names, if any, for each of the following compounds: (16.1, 16.4) CH3 a. CH3-CH-CH2-CH2-c-OH hey C- HO- b. Cl d. CH3-CH2–CH2-C-0–CH3 CH3 C-0-CH,-CH3 e. CH3-CH – CH2 CH3 OH f. CH3 CH- CH2 -СН-С-ОНarrow_forward
- (12.3)Which of the following has the strongest dispersion force between its molecules? O CH3CH3 O CH3CH₂CH₂CH₂CH3 O All of these have the dispersion forces with the same strength. O CH3CH₂CH₂CH3 O CH3CH₂CH₂CH₂CH₂CH3arrow_forward12.31 Write the IUPAC name for each of the following, using cis or trans prefixes, if needed: a. b. C. CH3-CH₂ H H H c=c C=C CH3 H H CH₂-CH₂-CH₂ - CH₂arrow_forward12.28 Draw the condensed structural or line-angle formula for the alkene produced by each of the following dehydration reactions: a. CH3 CH₂-OH b. C. OH OH Ht, heat Ht, heat H, heatarrow_forward
- 11.14 Give the IUPAC name for each of the following: CH3 a. CH3-CH-CH₂-CH₂-CH3 b. Br CH₂ - CH3 c. CH3-CH₂-CH-CH-CH₂-CH3 CH₂ CH3 d. -arrow_forward4.18 Write the IUPAC name for each compound: (See Examples 4.1, 4.5) a. b. C. d. е. f. 9. h.arrow_forward3.1 Write the IUPAC and common names for each of the following: a. CH3—CH₂—OH b. C. OH CH3-CH2-CH-CH3 SH ES d. OH $ OH Farrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY