
Concept explainers
(a)
Interpretation:
The three different sets of reagents (each set containing of a carbonyl compound and Grignard reagent) have to be used to prepare each of the following tertiary alcohols.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as
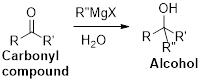
(b)
Interpretation:
The three different sets of reagents (each set containing of a carbonyl compound and Grignard reagent) have to be used to prepare each of the following tertiary alcohols.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as aldehyde or ketone, produces corresponding alcohol is the product and it is the one of the carbon – carbon bond forming reaction.
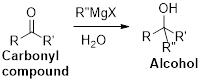
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
- Aldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forwardShow how each alcohol or diol can be prepared from an alkene. (a) 2-Pentanol (b) 1-Pentanol (c) 2-Methyl-2-pentanol (d) 2-Methyl-2-butanol (e) 3-Pentanol (f) 3-Ethyl-3-pentanol (g) 1,2-Hexanediolarrow_forwardThe following alcohol can be formed via two different Grignard reactions. Draw the GRIGNARD REAGENT which contains a BENZENE ring. Use the covalent format R-Mg-Br instead of the ionic format. Then draw the carbonyl compound Draw the GRIGNARD REAGENT. Use the covalent format R-Mg-Br instead of the ionic format. Then draw the carbonyl compound with a benzene ringarrow_forward
- Which of the following reagents would lead to a product that has two functional groups: an alcohol and an ether? NaOH/H₂O H3O+/H₂O HC1 CH3CH₂OH/CH3CH₂O™¹Na H₂C CH₂arrow_forwardName the following alcohol. O2-Bromo-4-ethylcyclopentanol 4-Ethyl-2-bromocyclopentanol O1-Ethyl-3-bromo-4-cyclopentanol O1-Bromo-4-ethyl-2-cyclopentanolarrow_forwardThe reaction of a ketone with an alcohol in acidic conditions will result in: an alkane an acetal a geminal (gem) diol an alkenearrow_forward
- When an aldehyde is treated with LiAlH4 followed by addition of H₂O, what general class of product results? ether secondary alcohol primary alcohol ketone tertiary alcoholarrow_forwardWhat is the IUPAC name for the following compound? O t-butyl cyclohexanecarboxylate O 3-cyclohexyl-2-oxy-tert-butanone O butyl cyclohexanone O t-butyl cyclohexanone O butyl cyclohexanecarboxylatearrow_forwardList three different sets of reagents (each set consisting of a carbonyl compound and a Grignard reagent) that could be used to prepare each of thefollowing tertiary alcohols:arrow_forward
- Rank the following alcohols in order of increasing ease of acid-catalyzed dehydration. Provide the structure of the dehydration product (alkene) from each alcohol. OH OH 3 1 a OHarrow_forwardWhat are the products formed when 1-butanal reacts with: NaOH/H2O CrO3, H2SO4, H2Oarrow_forwardThere are two possible Grignard reactions that lead to the given alcohol. Select the reactant needed for each of the indicated Grignard reagents. OH 1) ? MeMgBr 2) H3O* Harrow_forward

 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





