
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.49SP
The three isomers of dimethylbenzene are commonly named ortho-xylene, meta-xylene, and para-xylene. These three isomers are difficult to distinguish using proton NMR but they are instantly identifiable using 13 C N M R.
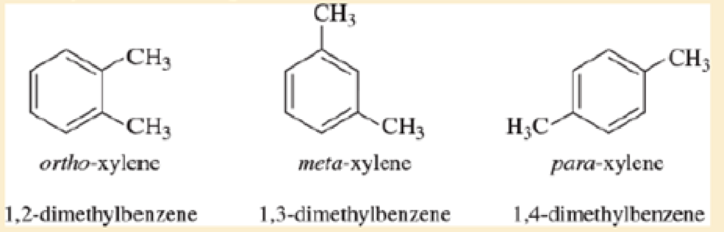
- a. Describe how carbon NMR distinguishes these three isomers.
- b. Explain why they are difficult to distinguish using proton NMR.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following molecules:
A.
H₂C
0
CH₂
C.
Ï_¶Ï
H₂C
B.
H₂C
A. acetone
C. ethanol
OH
B. methyl acetate
E. ethyl acetate
D. diethyl ether
is best represented by NMR spectra
below?
D.
20
H.C
17122
.CH₂₁
14
E.
H₂C
10
20
13C
2) The molecule corresponding to the NMR spectrum shown most likely
contains which of the following functional groups?
9
8
7
a. Aromatic ring
b. Ether
c. Aldehyde
d. Alkene
e. Alcohol
6
5
ppm
4
3
2
1
0
5. L-dopa is used in the treatment of Parkinson's disease. Identify the four most acidic
protons and arrange them in order of increasing acidity.
:O:
g
HO:
:OH
6. Coumarins are a class of natural products produced by plants and animals. It has a wide
variety of applications in cosmetics, food industry and fluorescent laser dyes. In the
¹HNMR spectrum of coumarin derivate shown below, the two signals farthest downfield
are at 7.38 and 8.42 pm. Identify the two protons that most likely give rise to these
signals and justify your answer using resonance structures.
:O:
Chapter 13 Solutions
Organic Chemistry (9th Edition)
Ch. 13.5A - In a 300-MHz spectrometer, the protons in...Ch. 13.5B - Prob. 13.2PCh. 13.6 - Determine the number of different kinds of protons...Ch. 13.6 - Prob. 13.4PCh. 13.7 - Draw the integral trace expected for the NMR...Ch. 13.7 - Prob. 13.6PCh. 13.8C - Draw the NMR spectra you would expect for the...Ch. 13.8D - Draw the NMR spectra you expect for the following...Ch. 13.8D - a. Assign protons to the peaks in the NMR spectrum...Ch. 13.8D - Prob. 13.10P
Ch. 13.8D - Two spectra are shown. Propose a structure that...Ch. 13.9 - Prob. 13.12PCh. 13.9 - The spectrum of trans-hex-2-enoic acid follows. a....Ch. 13.9 - Prob. 13.14PCh. 13.9 - Prob. 13.15PCh. 13.10 - Prob. 13.16PCh. 13.10 - If the imaginary replacement of either of two...Ch. 13.10 - Predict the theoretical number of different NMR...Ch. 13.11B - Prob. 13.19PCh. 13.11B - Prob. 13.20PCh. 13.11B - Prob. 13.21PCh. 13.11B - Prob. 13.22PCh. 13.11B - Prob. 13.23PCh. 13.11B - Prob. 13.24PCh. 13.12E - Draw the expected broadband-decoupled 13 C N M R...Ch. 13.12E - a. Show which carbon atoms correspond with which...Ch. 13.12E - Repeat Problem13-25, sketching the...Ch. 13.12F - Prob. 13.28PCh. 13.13 - A bottle of allyl bromide was found to contain a...Ch. 13.13 - A laboratory student was converting cyclohexanol...Ch. 13.14 - Sets of spectra are given for two compounds. For...Ch. 13 - An unknown compound has the molecular formula C 9...Ch. 13 - Prob. 13.34SPCh. 13 - Predict the approximate chemical shifts of the...Ch. 13 - Prob. 13.36SPCh. 13 - Prob. 13.37SPCh. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Prob. 13.40SPCh. 13 - For each compound shown below. 1. sketch the 13 C...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Prob. 13.44SPCh. 13 - Prob. 13.45SPCh. 13 - Prob. 13.46SPCh. 13 - A compound was isolated as a minor constituent in...Ch. 13 - Prob. 13.48SPCh. 13 - The three isomers of dimethylbenzene are commonly...Ch. 13 - a. Draw all six isomers of formula C 4 H 8...Ch. 13 - Prob. 13.51SPCh. 13 - Hexamethylbenzene undergoes free-radical...Ch. 13 - Each of these four structures has molecular...Ch. 13 - Prob. 13.54SPCh. 13 - Phenyl Grignard reagent adds to 2-methylpropanal...Ch. 13 - Prob. 13.56SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Given below are the ¹H NMR spectra of two isomeric compounds with the molecular formula C4H₂O₂. Spectrum X 3 C₂HgO₂ 11 Spectrum Y 10 9 8 0 11 10 ppm Using the following choices, determine which compound corresponds to which spectrum. LOCH 3 A. B. C. b D. C₂H₂O₂ مله 6 OH pom 3 6arrow_forward2. Rank Ha, Hb, and Hc in order of increasing chemical shift. Hb Hc H2 H3C CH3 Hd OCH2CH3 Hb Ha На Не а. b.arrow_forwardA signal is seen at 600 Hz from the TMS signal in an NMR spectrometer with a 300-MHz operating frequency. a. What is the chemical shift of the signal? b. What is its chemical shift in an instrument operating at 500 MHz? c. How many hertz from the TMS signal is the signal in a 500-MHz spectrometer?arrow_forward
- A'2 The 'H-NMR spectra of cyclohexanol and cyclohexanone are given below. Identify which spectrum belongs to which compound and assign the peaks in each spectrum that substantiate your decision.arrow_forward7. 100 80 Relative Intensity 8 20- 0 MS-NW-1589 25 50 75 100 m/z 125 c. Propose a structure for this compound. b. Calculate a possible molecular formula for this compound 150 a. This compound contains C, H, and one other atom. Identify the other atom from the mass spectrum and explain your reasoning. 175arrow_forwardTrying to figure out the structure based on this NMR. unsat=4 so probably includes benzene ring. Unsure of how substitued the ring is.arrow_forward
- 1. What is the number of signals that should be observed in the ¹H NMR spectra of each of these molecules? a. b. C. OH H₂N Text Textarrow_forwarda. b. identify the number of signals (i.e. types of hydrogen) for each structure below. predict the approximate chemical shift, integration (i.e. number of that type of hydrogen), and splitting for each signal in each structure. NO₂ NO₂ NO₂arrow_forward16. Predict the multiplicity for each shaded protons in the following compounds a. CH3CH₂CCl2CH3 C. CH3CH(CH3)CH3 b. CH3CH(CH3)OH d. CH3CH₂COOCH2CH3arrow_forward
- 3. For each compound give the number of NMR signals and then determine how many peaks are present for each NMR signal. а. b. С. Сarrow_forwardorgo chem II nmr please help me fill out this chart!arrow_forwardAnalyze the H1 NMR of the following reaction and label each Hydrogen. Then calculate the chemical shifts for each? Also write multiplicity?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY