
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 47P
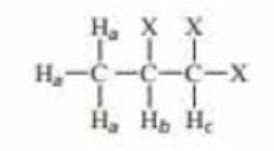
Draw a spitting diagram for the Hb proton and give its multiplicity if
- a. Jba = Jbc.
- b. Jba = 2Jbc.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a splitting diagram for the Hb proton if Jbc = 10 and Jba = 5.
Which of the following is R configuration?
...
OH
i
Br
(1)
a. (1)
b. (11)
C. (III)
€
(11)
O
d. (IV)
(111)
(IV)
a. Can a low-resolution mass spectrometer distinguish between C2H5+ and CHO+ ?b. Can a high-resolution mass spectrometer distinguish between them?
Chapter 15 Solutions
Organic Chemistry
Ch. 15.1 - Prob. 1PCh. 15.1 - Prob. 2PCh. 15.4 - How many signals would you expect to see in the 1H...Ch. 15.4 - Prob. 4PCh. 15.4 - How could you distinguish the 1H NMR spectra of...Ch. 15.4 - Draw an isomer of dichlorocyclopropane that gives...Ch. 15.5 - Prob. 7PCh. 15.5 - Prob. 8PCh. 15.5 - Prob. 9PCh. 15.5 - Where would you expect to find the 1H NMR signal...
Ch. 15.6 - Prob. 11PCh. 15.7 - Prob. 12PCh. 15.7 - Prob. 13PCh. 15.7 - Without referring to Table 14.1, label the proton...Ch. 15.8 - [18]-Annulene shows two signals in its 1H NMR...Ch. 15.9 - How would integration distinguish the 1H NMR...Ch. 15.9 - Which of the following compounds is responsible...Ch. 15.10 - Prob. 19PCh. 15.10 - Explain how the following compounds, each with the...Ch. 15.10 - The 1H NMR spectra of two carboxylic acids with...Ch. 15.11 - Draw a diagram like the one shown in Figure 14.12...Ch. 15.12 - Indicate the number of signals and the...Ch. 15.12 - How can their 1H NMR spectra distinguish the...Ch. 15.12 - Identify each compound from its molecular formula...Ch. 15.12 - Prob. 27PCh. 15.12 - Propose structures that are consistent with the...Ch. 15.12 - Describe the 1H NMR spectrum you would expect for...Ch. 15.13 - Prob. 30PCh. 15.13 - Identify the compound with molecular formula...Ch. 15.14 - Prob. 32PCh. 15.15 - a. For the following compounds, which pairs of...Ch. 15.17 - Explain why the chemical shift of the OH proton of...Ch. 15.17 - Prob. 37PCh. 15.17 - Prob. 38PCh. 15.17 - Prob. 39PCh. 15.20 - Answer the following questions for each compound:...Ch. 15.20 - Prob. 41PCh. 15.20 - How can 1,2-, 1,3-, and 1,4-dinitrobenzene be...Ch. 15.20 - Identify each compound below from its molecular...Ch. 15.22 - Prob. 44PCh. 15.22 - What does cross peak X in Figure 14.34 tell you?Ch. 15 - Prob. 46PCh. 15 - Draw a spitting diagram for the Hb proton and give...Ch. 15 - Label each set of chemically equivalent protons,...Ch. 15 - Match each of the 1H NMR spectra with one of the...Ch. 15 - Determine the ratios of the chemically...Ch. 15 - How can 1H NMR distinguish between the compounds...Ch. 15 - Prob. 52PCh. 15 - The 1H NMR spectra of three isomers with molecular...Ch. 15 - Prob. 54PCh. 15 - Prob. 55PCh. 15 - Prob. 56PCh. 15 - Compound A, with molecular formula C4H9Cl, shows...Ch. 15 - The 1H NMR spectra of three isomers with molecular...Ch. 15 - Would it be better to use 1H NMR or 13C NMR...Ch. 15 - There are four esters with molecular formula...Ch. 15 - An alkyl halide reacts with an alkoxide ion to...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Prob. 64PCh. 15 - How can the signals in the 6.5 to 8.1 ppm region...Ch. 15 - The 1H NMR spectra of two compounds, each with...Ch. 15 - Draw a splitting diagram for the Hb proton if Jbc...Ch. 15 - Sketch the following spectra that would be...Ch. 15 - How can 1H NMR be used to prove that the addition...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Dr. N. M. Arr was called in to help analyze the 1H...Ch. 15 - Calculate the amount of energy (in calories)...Ch. 15 - The following 1H NMR spectra are four compounds,...Ch. 15 - When compound A (C5H12O) is treated with HBr, it...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Identify each of the following compounds from its...Ch. 15 - Identity the compound with molecular formula...Ch. 15 - Identify the compound with molecular formula C6H14...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the Ka value for each of the compounds? Express the answers in proper scientific notation. A. Determine the Ka value for the compound with pKa = 10.26. H3C- OH B. Determine the K₂ value for the compound with pK₂ = 16.50. OH C. Determine the K₂ value for the compound with pKa = 5.21. K₁ = K₂ = K₁ =arrow_forward(iii) H,O2 + Н-Br (iv) (1) O3 + (ii) Zn/H,Oarrow_forwardWhy is it important for solutions to be diluted before they are run through spectroscopy?arrow_forward
- In mass spectrometry, the molecular ion peak occurs at O A. the largest m/z value. O B. the smallest m/z value. O C. the m/z value of the most abundant ion. O D. an m/z value equal to the molecular weight of the sample molecule.arrow_forwardWhich of the indicated protons would have the smallest pKa value? C. H. H CH3 EC H H. d. b. b. C A Moving to another question will save this response. O O O 0arrow_forward12. Which absorption spectrum corresponds to a red solution? a. (a) b. (b) C. (c) d. (a) and (c) e. (b) and (c) 400 nm e 700 nmarrow_forward
- The leaves of the Brazilian Tree Senna multijunga contain a number of pryidine alkaloids that inhibit acetylcholinterinase. Two recentyl isolated isomeric compounds have the strcture have the strcture shown below. (NOTE: M=293) Use the mass spectral data provided to determine the precise location of the hydroxyl group in each isomer. Isomer A: EI-MS, m/z(rel. int): 222(20), 150(10), 136(25), 123(100) Isomer B:EI-MS, m/z(re;. int): 236(20), 150(10), 136(25), 123(100)arrow_forwardIf you were performing following reaction (5) in the lab, how could you use IR spectroscopy to determine if the reaction worked? SELECT THE ANSWER FROM THE FOLLOWING THREE OPTIONS: A) Formation of C-Br bond and loss of C=C bond B) Loss of C-Br bond and appearance of C-OMe bond C) Loss of C-Br bond and appearance of C=C bondarrow_forward7. Given the pK, information for different acids below, place the following acids in order of increasing acid strength (from weakest to strongest). Anilinium ion, CSH6N*, pKa = 4.60 Benzilic acid, pka = 3.09 %3D Chloroacetic acid, pKa = 2.98 %3D Dibromophenol, pka = 8.06 %3D a) Anilinium ion → Benzilic acid → Chloroacetic acid → Chloroacetic acid b) Dibromophenol → Chloroacetic acid → Benzilic acid → Anilinium ion c) Chloroacetic acid → Benzilic acid → Anilinium ion → Dibromophenol d) Dibromophenol → Anilinium ion → Benzilic acid → Chloroacetic acid e) Chloroacetic acid → Anilinium ion → Dibromophenol→ Benzilic acid f) none of the abovearrow_forward
- Predict the ¹H shifts of the protons on the labeled carbons of the following chemical structure: OH NH₂ 1 2 a. H(1)=6.44, H(2)=6.40 b. H(1)=6.64, H(2)=6.60 c. H(1)-5.46, H(2)=5.40arrow_forward1. Which one of the given compounds is consistent with the mass spectrum below? 100 40 20 10 20 25 30 35 40 5o 55 45 60 65 70 75 m/z Courtesy of SDBS: National Institute of Advanced Industrial Science and Technology A. CH,CH,CH(CH,), В. CH,CHOHCH,CH, C. CH,CH,OCH,CH, D. CH,CH,NHCH,CH, E. CH,CH,CH,CH, Relative htensityarrow_forwardCan a low-resolution mass spectrometer distinguish between C2H5+ and CHO+? a. Can a high-resolution mass spectrometer distinguish between them?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY