
Concept explainers
A step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring.
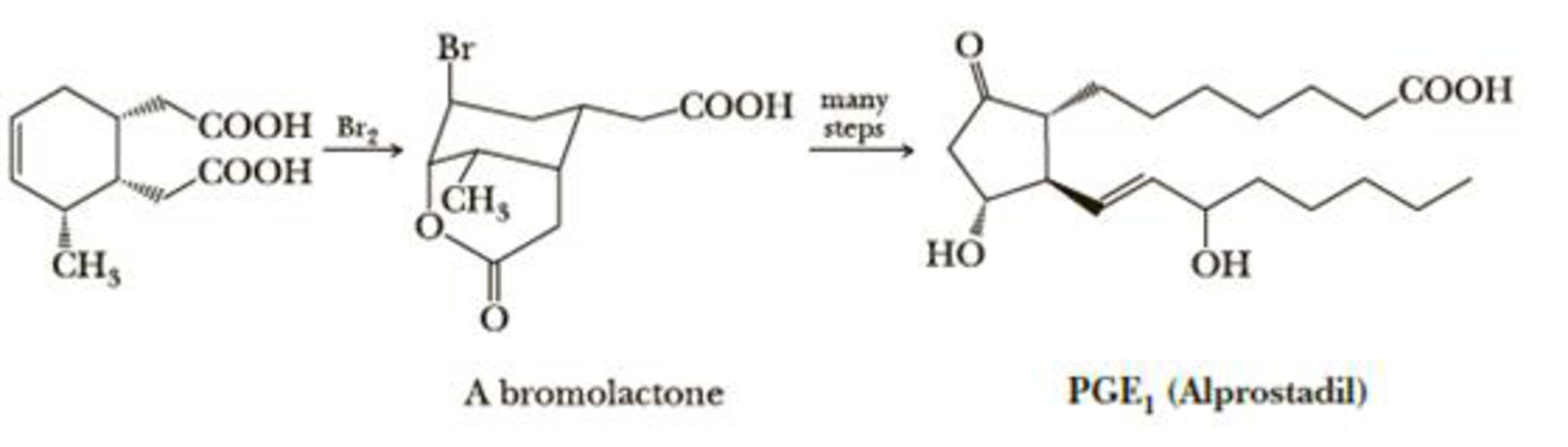
Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.
Trending nowThis is a popular solution!

Chapter 18 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry
Organic Chemistry - Standalone book
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: The Molecular Nature of Matter
EBK INTRODUCTION TO CHEMISTRY
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardFollowing is a retrosynthetic analysis for an intermediate in the industrial synthesis of vitamin A. (a) Addition of one mole of HCl to isoprene gives 4-chloro-2-methyl-2-butene as the major product. Propose a mechanism for this addition and account for its regioselectivity. (b) Propose a synthesis of the vitamin A precursor from this allylic chloride and ethyl acetoacetate.arrow_forwardWhen cis-4-chlorocyclohexanol is treated with sodium hydroxide in ethanol, it gives mainly the substitution product trans-1,4-cyclohexanediol (1). Under the same reaction conditions, trans-4-chlorocyclohexanol gives 3-cyclohexenol (2) and the bicyclic ether (3). (a) Propose a mechanism for formation of product (1), and account for its configuration. (b) Propose a mechanism for formation of product (2). (c) Account for the fact that the bicyclic ether (3) is formed from the trans isomer but not from the cis isomer.arrow_forward
- Identify products A and B from the given 1H NMR data. Treatment of CH2=CHCOCH3 with one equivalent of HCl forms compound A. A exhibits the following absorptions in its 1H NMR spectrum: 2.2 (singlet, 3H), 3.05 (triplet, 2 H), and 3.6 (triplet, 2 H) ppm. What is the structure of A?arrow_forwardTreatment of cis-4-bromocyclohexanol with HO– affords compound A and cyclohex-3-en-1-ol. Treatment of trans-4- bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and B contain different functional groups and are not isomers of each other. Propose structures for A and B and offer an explanation for their formation.arrow_forwardWhen cis-2-decalone is dissolved in ether containing a trace of HCI, an equilibrium is established with trans-2-decalone. The latter ketone predominates in the equilibrium mixture. H H HCI cis-2-Decalone trans-2-Decalone Propose a mechanism for this isomerization and account for the fact that the trans iso- mer predominates at equilibrium.arrow_forward
- The UV spectrum of 1-phenylprop-2-en-1-ol shows an intense absorption at 220 nm and a weaker absorption at 258 nm. When this compound is treated with dilute sulfuric acid, it rearranges to an isomer with an intense absorption at 250 nm and a weaker absorption at 290 nm. Suggest a structure for the isomeric product and propose a mechanism for its formation.arrow_forwardAn alkene is treated with OsO4 followed by H2O2. When the resulting diol is treated with HIO4, the only product obtained is an unsubstituted cyclic ketone with molecular formula C6H10O. What is the structure of the alkene?arrow_forwardFollowing is the structural formula of Surfynol, a defoaming surfactant. Describe the synthesis of this compound from acetylene and a ketone. How many stereoisomers are possible for Surfynol? OH OH Surfynolarrow_forward
- Describe how 3-methyl-1-phenyl-3-pentanol can be prepared from benzene. You can use any inorganic reagents and solvents, and any organic reagents provided they contain no more than two carbons.arrow_forwardIdentify A, B, and C, intermediates in the synthesis of the five-membered ring called an α- methylene-γ-butyrolactone. This heterocyclic ring system is present in some antitumor agents.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

