
Concept explainers
A phase diagram for carbon and platinum is shown. It is assumed the lines shown are linear, and the mixture has the following characteristics. The endpoints of the division line between these two phases are labeled on the diagram.
■ Below 1700 degrees Celsius, it is a mixture of solid platinum (Pt) and graphite (G).
■ Above 1700 degrees Celsius, there are two possible phases: a Liquid (L) phase and a Liquid (L) + phase (G).
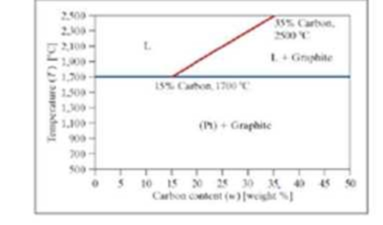
Write a program to determine the phase. The program should ask the user to enter the weight percent carbon and the temperature in degrees Celsius. Call the phases “Pt + G,” “L,” and “L + G,” for simplicity. If the point falls directly on the T = 1700°C line, include the point in the “Pt + G” phase. If the point falls directly on the L, L + Gline, include the point in the “L + G” phase.
Store the phase as text in a variable. The equation of the line dividing the L and L + G phases must be found in the program using polyfit. The program should produce a formatted output statement to the command window. Similar to “For X.XX weight percent carbon and a temperature of YYY degrees Celsius, the phase is PHASE” where x.xx, yyy and PHASE are replaced by the actual values formatted as shown.
The following partial code is designed to implement this program. You are to fill in the missing sections of code as appropriate to complete the program.

Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
- You are working with a derivative of naphthalene and, like its parent compound, it has a high triple point and sublimates fairly readily at temperatures near room temperature. At temperatures below 400 K, you find that the vapor pressure of its solid can be represented by the function P 4200 K (1)-11.5 = 11.5 1 atm T Use this equation to estimate the normal sublimation temperature of this compound, Tsubl (in K).arrow_forwardFor the metals A and B, which are given below, create the hypothetical phase diagram between 500 ° C and 900 ° C temperatures.Ø A metal has a melting temperature of 840 ° C.The solubility of Ø B in A can be ignored at all temperatures.Ø Melting temperature of Metal B is 730 ° C.The maximum solubility of A in Ø B is 10% A by weight, which occurs at 600 ° C.At 500 ° C, the solubility of A in B is 6% A by weight.Ø An eutectic 600 ° C and 74% B by weight - 26% by weight A are formed.Ø The second eutectic occurs at 630 ° C and 59% B - 41% A by weight.Ø A third eutectic occurs at 655 ° C and 39% B - 61% A by weight.Ø A congress melting point is formed at 680 ° C and 52% B - 48% A by weight.Ø A second congress melting point is formed at 655 ° C and 66% B - 34% A by weight.Ø Intermetallic compound AB is present at 52 wt% B - 48 wt% A.Ø Intermetallic compound AB2 is present at 66 wt% B to 34 wt% A.arrow_forwardProblem (Separation of a Mixture of Benzene and Toluene) A 100 kgh mixture of benzene (B) and toluene (T) containing 50% benzene by mass is separated by distillation into two fractions. The mass flow rate of benzene in the top stream is 45 kg B/h and that of toluene in the bottom stream is 47.5 kg Th. The operation is under steady state.Write balances for benzene and toluene to calculate the unknown componentflow rate in the output streams.arrow_forward
- The phase diagram for sulfur is shown here. The rhombic and monoclinic states are two solid states with different structures. a. Below what pressure does solid sulfur sublime? b. Which of the two solid states of sulfur is more dense? Liquid Rhombic Monoclinic 119 °C, 0.027 mmHg 96 °C, 0.0043 mmHg Vapor Temperature Pressurearrow_forwardThe following question refer to the phase diagram shown here. Composition (at% Sn) 60 T Temperature (°C) 300 200 OC 0 100 327°C O 98 wt% Sn O 100 % O 61.9 wt% Sn 0 0% O 73 wt% Sn 20 T O 50% O 90 wt% Sn 18.3 40 T 20 183°C a + ß 40 T Liquid 61.9 80 B+L 80 232°C 97.8 100 →600 0 100 60 Composition (wt% Sn) (Pb) (Sn) Adapted from Binary Alloy Phase Diagrams, 2nd edition, Vol. 3, T. B. Massalski (Editor-in-Chief), 1990. Reprinted by permission of ASM International, Materials Park, OH. In the Pb-Sn phase diagram, what is the approximate composition of the 3 phase for an alloy with 90 wt% Sn and at 200°C? 500 400 B 300 a 200 Temperature (°F) 100arrow_forwardAssignment 4 Eхample A gas mixture has the following composition by volume: 20% N, 30% CO, 50% CH. Other data: For N, C,1 = 1.0414; CO, C = 0.8452; CH,, C = 2.1377 → kJ/kg K Find for the mixture (a) molecular weight (b) gas constant (c) gravimetric analysis (% by mass) (d) k %3Darrow_forward
- 100 grams of water are mixed with 200 grams of another fluid (specific density=790 kg/m3), what is the specific volume of the resulting mixture, assuming the volumes are additive and the mixture is homogenous?arrow_forwardFigure 1 shows a binary phase diagram of elements A and B. Determine the phases present, composition, and amount of each phase at 215 °C. T(°C) 300+ 215 200+ 100+ 0 A L+a 1720 Ca L (liquid) 183°C a + ß 40 46 60 Co CL wt% 80 +B 100 Barrow_forwardi) Draw the phase diagram of a binary A (850 °C) and B (870 °C) system using below given information. • A and B are completely soluble in liquid phase. • B has no solid solubility in A. • Solid solubility of A in B at room temperature is 5 % and its maximum value is 20 %. • Eutectic point: 55 % B; 550 °C ii) for an alloy for which 55% Barrow_forward1. Immiscible components do not mix in the liquid phase, and each independently exerts its saturation pressure until they rach a certain phase they can form homogenous mixture? 2. It will be easier to separate the components of a vapor micture if they have the same concentration. True or Falsearrow_forwardA system consists of 50% mole percent of propane, 20% mole percent of pentane, and 30% mole percent of heptane at 150°F. Detail as shown in Table 2 below. Assuming ideal solution behaviour and using the Raoult's Law (where x₁ = mole fraction in liquid phase), Table 2: Gaseous properties Gas Molecular weight (g/mol) 44.1 Xi 72.15 100.21 Propane Pentane Heptane (i) Calculate the total vapor pressure (P+) of the system. 0.5 0.2 0.3 Pvap at 150°F (psia) 345.0 36.6 50 (ii) Calculate the gas/vapor phase specific gravity. Given the SGair is 28.96 g/mol. (iii) Calculate the liquid phase specific gravity. Given the SGwater is 62.4 lb/ft³.arrow_forward100 atm 1 atm 0.118 atm 114 °C 184 °C 535 °C Temperature (not to scale) a.) Use the above generic phase diagram, clearly identify where you would find the following: Gas, Liquid, Solid, Triple Point, Draw arrows and label to indicate the six transitions (melting, freezing, sublimation, deposition, vaporization and condensation) b.) Based on the above phase diagram, what phase would you be in at a pressure of 50 atm and 300°C? c.) Based on the above phase diagram at what temperature in °C would vaporization occur under normal conditions? d.) If the pressure was 0.050 atm and 425°C, what phase would you be in? e.) If the pressure was 0.118 atm and temperature was 114°C, what phase(s) would you be in? f.) At approximately what temperature would the normal freezing point be in °C? g.) If the pressure was 0.105 atm and starting at a temperature of 32°C to 450°C what phase changes would occur (put in increasing temperature order). Pressure (not to scale)arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





