
Concept explainers
a.
Interpretation: The major form of alanine present at
Concept Introduction: Amino acids are compounds that contain both an amino group and a carboxyl group. Alanine is an amino acid having molecular formula,
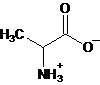
b.
Interpretation: The major form of alanine present at
Concept Introduction: Amino acids are compounds that contain both an amino group and a carboxyl group. Alanine is an amino acid having molecular formula,
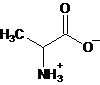
c.
Interpretation: The major form of alanine present at
Concept Introduction: Amino acids are compounds that contain both an amino group and a carboxyl group. Alanine is an amino acid having molecular formula,
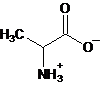
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Introduction To General, Organic, And Biochemistry
- Answer the following statement. Provide an explanation. What happens to compound X when ph=pkb ph>pkb pharrow_forward1a) Consider the reaction AH(+) + H₂O A: + H3O+). For the following named acids: a) draw the structure of the acid, b) give the approximate pKa of the conjugate acid in units of 5, c) give the name of the conjugate base, and d) draw the structure of the conjugate base. p-Toluenesulfonic acid acid Conjugate base Imidazolium cation pka_ pKa iso-Propanol pKa Acetylene pKaarrow_forwardPhenylacetic acid (C₂H₂CH₂COOH) has a K₂ of 4.9 x a 10-5. Calculate for the pH, pOH, [H+] and [OH-] of a solution of phenylacetic acid that is 0.186 M.arrow_forward
- Name two techniques that can be employed to extract oil from seeds Hence,Name four processes that may be used to refine oils,What is the description for the term rancidity of oils? ,Name two causes of rancidity in oils, What is acid value of oils? Therefore How many moles of KOH is required per mole of fat in saponification reactions?arrow_forward(a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forwardYou are looking at extracting a pyridine-bearing compound (conjugate acid pKA-4) from an organic phase and transfer it to the aqueous phase. Which of the following aqueous solutions can you use to do so? IM NGOH,pH 14 water, pH 7 saturated NH4Cl,pH 5 1MHCl, pH 1arrow_forward
- Each molecule or ion can function as a base. Write a structural formula of the conjugate acid formed by reaction of each with HCl. Q.) CH3CH2OHarrow_forward2. Will a 0.10 molar solution of CH3NH3OCN (methylamine cyanate) produce a basic or acidic solution. Explain quantitatively.arrow_forwardIn the following reaction in aqueous solution, the base reactant is and its conjugate acid product is CH3COOH(aq) + NH3(aq) CH3COO (ag) + NH4 (ag) O NH3: NH4 NH3: CH3COO CH3COOH; NH4 CH3COOH; H30+ CH3COOH; CH3CO*arrow_forward
- Complete the structures of these two am ino acids at the pH values given. Explain why the structures change at different pH values. ÇH3 H2N- -COOH H2N-C СООН H H alanine glycine at pH 1 at pH 7 at pH 14arrow_forwardRank the Acidity of the following sets of compounds. Give an explanation for the answers.arrow_forwardWhat is the conjugate acid of each of the following? What is the conjugate Dase of eacn? (a) NH Parts of the formula in your answer are transposed. Your answer appears to be missing a charge. Conjugate acid (b ) (CH₂)₂NH \table[[chemPad, (9) Help], [xlx =larr|l, Greek -],[(CH),NH,N What is the conjugate acid of each of the following? What is the conjugate base or eacn (UTIL SOLES- (a) NH3 Conjugate acid Conjugate base (b) (CH3)2NH Conjugate acid Conjugate base chemPad XX→→ chemPad XX→ NH₂ NH 2 NH4 NH 4 Parts of the formula in your answer are transposed. Your answer appears to be missing a charge. chemPad XX→ (CH4)2NH2 (CH_4)_2NH_2 chemPad XX→= Greek (CH₂)2N (CH_2) 2N Help Greek Help Greek X Help Greek juur Helparrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

