
Concept explainers
a)
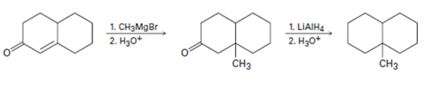
Interpretation:
The flaw in the reaction scheme given is to be identified and the corrected scheme is to be provided.
Concept introduction:
α,β-Unsaturated
To identify:
The flaw in the reaction scheme given and to provide the corrected scheme.
b)

Interpretation:
The flaw in the reaction scheme given is to be identified and corrected scheme is to be given.
Concept introduction:
CrO3 in the presence of H3O+ oxidize primary alcohols to acids and not to
To identify:
The flaw in the reaction scheme given and to provide the corrected scheme.
c)
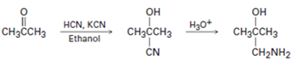
Interpretation:
The flaw in the reaction scheme given is to be identified and corrected scheme is to be given.
Concept introduction:
Aldehydes and ketones react with HCN, KCN to yield the corresponding cyanohydrins. The cyanohydrins when treated with aqueous acids will get hydrolysed to acids.
To identify:
The flaw in the reaction scheme given and to provide the corrected scheme.
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
- In each reaction box, place the best reagent and conditions from the list provided. 1) Br 2) 3) 4) DEET (the active ingredient in over the counter insect repellant) 5) Answer Bank Mg, Et,0 HN(CH, CH, ), (1 equiv.) H,O* CH,COOH CH,CH,NH, CH,CH, Br NH(CH,CH,),(2 cquiv.) НСООСH, H,CO НСООН CH, CH, OH NaCN NaNH, Br,, FeBr, SOCI, CO,arrow_forwardSECTION 6: Show the mechanism of each reactions with arrows and intermediates form as in a. b. C. HBr Br₂ Cl₂, H₂O Br Br CI Br OHarrow_forward7. Reaction Scheme. о А N 1. xs Mel, xs K2CO3 2. Ag2O, H₂O 3. heat NH2 NH2 or two differnet methods (no same steps/reagents) C5H12N2 Br2, xs NaOH, xs H₂O OHC 1.03 2. DMS CHOarrow_forward
- Is the following true or false? A highly selective reaction is one that proceeds with a high turn-over number.arrow_forwardFrom the reaction conditions provided, design a four-step reaction sequence that is expected to convert the starting material into the product shown? For each step, only the major expected product can be taken forward. Assume standard workup as necessary. of 1. Cros, pyridine 2. BHs, THF, H2O2, H20 3. HBr 4. (COC2)2. MeOH 5. HBr, ROOR, heat 6. NBS, light 7. tBuONa, tBuOH 8. H2SO4, H2O Step 1: choose your answer. Step 2: choose your answer. v Step 3: choose your answer. Step 4: >arrow_forward7. The reaction of methoxide anion with bromoethane to yield the ether ethyl methyl ether and the bromide anon (Br-) is an excellent example of a general reaction type called Sy2 (substitution nucleophilic bimolecular): CH,0+ CH,СH-Br a CH3-0-CH,СH; + Br- a. Change in enthalpy is -103 kJ/mol; the change in entropy is + 0.025 kJ/mol-K. Calculate DG at 300K. b. Is the reaction endergonic or exergonic? c. Is the reaction endothermic or exothermic? d. Use curved arrows to show the complete mechanism. Reaction of 2-methyl-1-butene with H-Cl could yield TWO alkyl chloride products. Draw and name 8. them.arrow_forward
- Which of the following synthetic routes is the one that will most successfully generate a. 1. Brz. CH,COOH CH,CH,CH,C-OEt CH,CH,CH=CHC-OEt 2. Pyridine, heat b. ÇH, CH,CH,C-COOH 1. Na* OEt CH,(CO,Et), 2. CH,CH,Br 3. Na* OEt 4. CH,Br 5. H,O*, heat с. 1. Na* OEt CH,CH,C-OEt H,C=CHCH,CH,COOH 2. Н.С-СHCH, Br 3. Н, о", heat d. ÇO̟Et ÇO̟,Et ÇH, -CHCOOH 1. Na**OEt CH,CH-CO,Et 2. PhBr 3. H,O*, heatarrow_forwardFrom the reaction conditions provided, design a four-step reaction sequence that is expected to convert the starting material into the product shown? For each step, only the major expected product can be taken forward. Assume standard workup as necessary. 1. HBr, ROOR, heat 2. NBS, light 3. tBuONa, tBuOH 4. H2SO4, H2O 5. (COCI2)2, MeOH 6. ВНз, ТHF, H202, Н2О 7. HBr 8. CrOз, рyridine Step 1: choose your answer... Step 2: choose your answer... Step 3: choose your answer... Step 4: choose your answer.. Nextarrow_forwardFill in the blanks of the following reaction scheme using any reasonable starting materials and reagents. What is the first reagent used in the second reaction (i.e., "1. ")? H+ он NABH4 NaCN O 1. NaCN, HCI, H2O, heat H2SO4, H2O О (СН3)2CНОН (еxcess) LIAIH4arrow_forward
- (1) MeO₂C CO₂Me (ii) (iii) H H₂C 1. Reagents 2. Reaction Conditions 3. Mechanism 1. Reagents 2. Reaction Conditions 3. Mechanism 1. Reagents 2. Reaction Conditions 3. Mechanism H₂C CH3 O OH OH Afla H₂C OMearrow_forwardComplete the following reaction schemes:arrow_forward3. For questions "A"- "C", complete the following reaction schemes (no mechanisms required). Show all reagents used in the order of application. th A) 6 B) ||| Ins HO HOarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

