
Concept explainers
(a)
Interpretation:
The condensed structural formula has to be drawn for the given
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
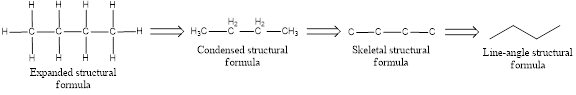
(b)
Interpretation:
The condensed structural formula has to be drawn for the given unsaturated hydrocarbon.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
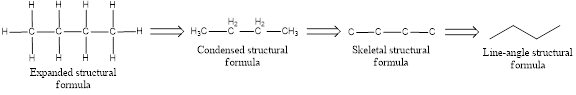
(c)
Interpretation:
The condensed structural formula has to be drawn for the given unsaturated hydrocarbon.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
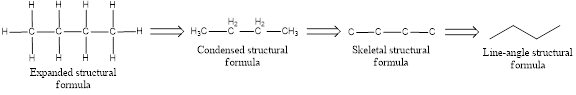
(d)
Interpretation:
The condensed structural formula has to be drawn for the given unsaturated hydrocarbon.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
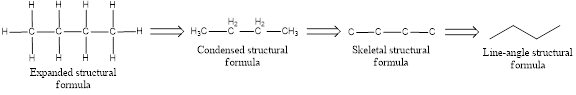
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic And Biological Chemistry
- What is the difference in bonding and in the general molecular formula between an alkene and an alkane with the same number of carbon atoms?arrow_forwardDraw the structure of each alkane and cycloalkane from the given incorrect name. Then, give the IUPAC name for each compound. a. 7-ethyl-3,6-dimethylnonane b. 4-ethyl-3-isopropylheptane c.3-ethyl-1,4-dimethylcycloheptane d. 1-ethyl-3-methyl-5-isopropylcyclohexanearrow_forwardWhich of the following can exhibit geometric isomerism? a. 1-propene b. 1,2,2-tribromoethene c. 2,3-dimethyl-2-butene d. 1-bromo-1-propenearrow_forward
- Write a balanced equation for the hydrobromination of each of the following alkenes.a. 2-Pentene c. 3-Hepteneb. Propene d. Ethenearrow_forwardWhich of the following is a CORRECT name according to the IUPAC rules? a. 2-ethyl-2-methylpentane b. 3,4-dimethylpentane c. 3-ethyl-2-methylpentane d. 2-methylcyclohexanearrow_forward4. The following names are for actual compounds, but the name given are incorrect. Draw out the structures and give the proper IUPAC name. a. 4-ethylpentane b. 2-ethyl-3-methylpentane c. 2,2-diethylheptane d. 2-propylpentane e. 4,4-diethylbutanearrow_forward
- 2. Draw structures corresponding to the following IUPAC names. a. 2-Methyl-1,5-hexadiene b. 3-Ethyl-2,2-dimethyl-3-heptene c. 2,3,3-Trimethyl-1,4,6-octatriene d. 3,4-Diisopropyl-2,5-dimethyl-3-hexene e. 3-methyl-1-pentyne f. (Z)-3-methylhex-2-en-4-ynearrow_forwardExplain why each of the following is an incorrect IUPAC name. Write the correct IUPAC name for the compound. a. 1,3-Dimethylbutane b. 4-Methylpentane c. 2,2-Diethylbutane d. 2-Ethyl-3-methylpentane e. 2-Propylpentane f. 2,2-Diethylheptane g. 2,2-Dimethylcyclopropane h. l-Ethyl-5-methylcyclohexanearrow_forward13. Draw condensed structural diagrams and line structural diagrams for the following alkene names: a. 3-methylbut-1-ene b. 3-ethyl-4-methyl-3-propylhept-1-ene c. 2-ethyl-3,4-dimethylhept-1-ene d. 3-methylpent-1-ene e. 3-ethyl-4,5-dimethylhex-1-enearrow_forward
- Which of the following exhibits cis/trans isomerism a. 1-butene b. 2,3-dimethyl-2-butene c. 2-butene d. propenearrow_forwardDraw the carbon skeletal structure for the following organic compounds and identify the main functional group or family. A. 1-methyl 1-cyclopentanol B. 3,4-diethyl nonanal C. 2,3,4,4-tetrabromo-1-octene D. 3-methyl pentanoic acid E. ethyl dimethylaminearrow_forwardThe correct IUPAC name for the following compound is CH2 H2 H3C. CH3 H2 H3C CH3 a. 2,3,3-trimethyl-1-hexene b. 2,3,3-trimethylhexane c. 4,4,5-trimethylhexane d. 3,4,4-trimethyl-2-hexenearrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



