
Concept explainers
(a)
The temperature of the gas at B if the temperature at A is
(a)
Answer to Problem 51SP
Solution:
Explanation of Solution
Given data:
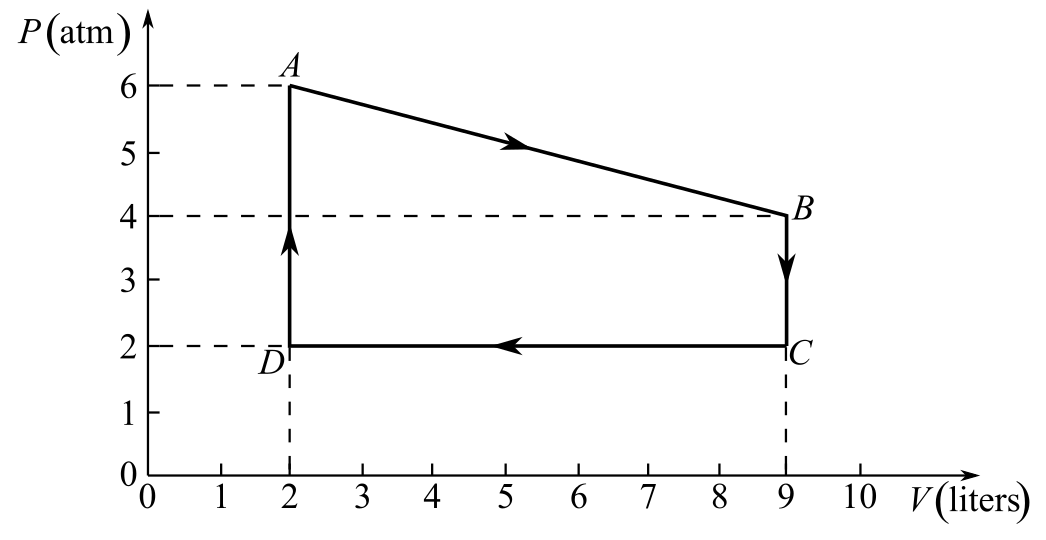
Refer to Fig. 20-6.
The temperature at A is
The mass of gas enclosed in the cylinder is
The gas follows the process A to B in the thermodynamic cycle shown in Fig. 20-6.
Formula used:
The gas equation for a process is expressed as
Here,
The formula for the conversion of the initial temperature of a gas from the Celsius scale to the Kelvin scale is
Here,
Explanation:
Draw the thermodynamic cycle diagram given in Fig- 20.6:
Recall the expression for the conversion of temperature at A from Celsius to Kelvin:
Here,
Substitute
Refer to the diagram and write the values of pressure and volume at points A and B, respectively,
And
Here,
Recall the gas equation between points A and B:
Here,
Substitute
Conclusion:
The temperature at point B is
(b)
The value of
(b)
Answer to Problem 51SP
Solution:
Explanation of Solution
Given data:
Refer to Fig. 20-6.
The temperature at A is
The heat received by the gas from A to B is
The mass of the gas enclosed in the cylinder is
Formula used:
The area of a trapezium is calculated by the formula:
Here,
The work done in a thermodynamic process is given by the area under the line representing the process in the pressure–volume diagram:
Here,
The first law of thermodynamics for a process is written as
Here,
The formula for change in internal energy is
Here,
The formula for conversion of temperature of gas from Kelvin scale to Celsius scale is
Here,
Explanation:
Draw the thermodynamic cycle diagram given in Fig- 20.6:

Understand that the work done in the thermodynamic process AB is equal to the area of the pressure–volume diagram under the line AB.
Draw the thermodynamic cycle diagram showing the area under the line AB:
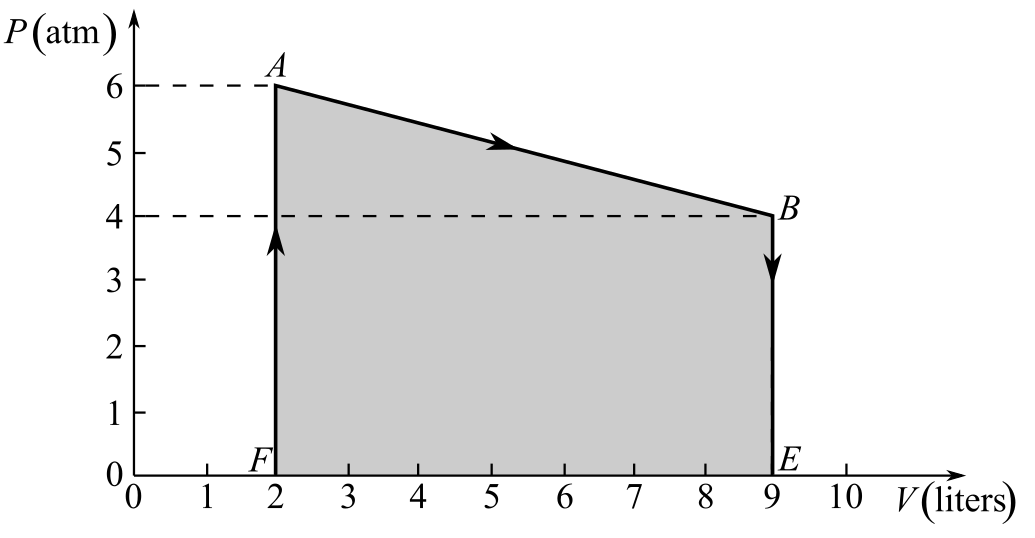
Here, the points E and F are as shown in the figure and the work done during the process AB is represented by the area under the line AB, which is equal to the area of the trapezium ABEF.
Refer to the figure and write the values of the lengths of sides AF, BE, and EF:
Recall the expression for the area of trapezium ABEF to calculate the area under the line AB in order to calculate the work done in the process AB:
Here,
Substitute
Recall the expression for the net-work done in a thermodynamic process in terms of the area of the pressure–volume diagram:
Substitute
Recall the expression for the first law of thermodynamics for the process AB:
According to the problem, the heat supplied from A to B is
Substitute
Further solve as
Calculate the temperature at point B in Celsius:
Here,
Substitute
Calculate the change in temperature from A to B:
Substitute
Recall the formula for change in internal energy:
Substitute
Conclusion:
The value of
Want to see more full solutions like this?
Chapter 20 Solutions
Schaum's Outline of College Physics, Twelfth Edition (Schaum's Outlines)
- A car tile contains 0.0380 m3 of air at a pressure of 2.20105 Pa (about 32 psi). How much more internal energy does this gas have than the same volume has at zero gauge pressure (which is equivalent to normal atmospheric pressure)?arrow_forwardOne of a dilute diatomic gas occupying a volume of 10.00 L expands against a constant pressure of 2.000 atm when it is slowly heated. If the temperature of the gas rises by 10.00 K and 400.0 J of heat are added in the process, what is its final volume?arrow_forwardTwo moles of a monatomic ideal gas at (5 MPa, 5 L) is expanded isothermally until the volume is doubled (step 1). Then it is cooled isochorically until the pressure is 1 MPa (step 2). The temperature drops in this process. The gas is now compressed isothermally until its volume is back to 5 L, but its pressure is now 2 MPa (step 3). Finally, the gas is heated isochorically to return to the initial state (step 4). (a) Draw the four pi-cresses in the pV plane. (b) Find the total work done by the gas.arrow_forward
- The excess internal energy of metabolism is exhausted through a variety of channels, such as through radiation and evaporation of perspiration. Consider another pathway for energy loss: moisture in exhaled breath. Suppose you breathe out 22.0 breaths per minute, each with a volume of 0.600 L. Suppose also that you inhale dry air and exhale air at 37C containing water vapor with a vapor pressure of 3.20 kPa. The vapor comes from the evaporation of liquid water in your body. Model the water vapor as an ideal gas. Assume its latent heat of evaporation at 37C is the same as its heat of vaporization at 100.C. Calculate the rate at which you lose energy by exhaling humid air.arrow_forwardIf a gas is compressed isothermally, which of the following statements is true? (a) Energy is transferred into the gas by heat. (b) No work is done on the gas. (c) The temperature of the gas increases, (d) The internal energy of the gas remains constant, (e) None of those statements is true.arrow_forwardAn amount of n moles of a monatomic ideal gas in a conducting container with a movable piston is placed in a large thermal heat bath at temperature T1 and the gas is allowed to come to equilibrium. After the equilibrium is leached, the pressure on the piston is lowered so that the gas expands at constant temperature. The process is continued quasi-statically until the final pressure is 4/3 of the initial pressure p1 . (a) Find the change in the internal energy of the gas. (b) Find the work done by the gas. (c) Find the heat exchanged by the gas, and indicate, whether the gas takes in or gives up heat.arrow_forward
- A sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forwardOne process for decaffeinating coffee uses carbon dioxide ( M=44.0 g/mol) at a molar density of about 14,0 mol/m3 and a temperature of about 60 . (a) Is CO2 a solid, liquid, gas, or supercritical fluid under those conditions? (b) The van der Waals constants for carbon dioxide are a=0.3658 Pa m6/mol2 and b=4.286105 m3/mol. Using the van der Waals equation, estimate pressure of CO2 at that temperature and density. `arrow_forwardAn aluminum rod 0.500 m in length and with a cross-sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 300 K. (a) If one-half of the rod is inserted into the helium, how many liters of helium boil off by the time the inserted half cools to 4.20 K? Assume the upper half does not yet cool. (b) If the circular surface of the upper end of the rod is maintained at 300 K, what is the approximate boil-off rate of liquid helium in liters per second after the lower half has reached 4.20 K? (Aluminum has thermal conductivity of 3 100 W/m K at 4.20 K; ignore its temperature variation. The density of liquid helium is 125 kg/m3.)arrow_forward
- An ideal gas has a pressure of 0.50 atm and a volume of 10 L. It is compressed adiabatically and quasi-statically until its pressure is 3.0 atm and its volume is 2.8 L. Is the monatomic, diatomic, or polyatomic?arrow_forwardOne might think that the internal energy of diatomic gases is given by Eint=5RT/2 . Do diatomic gases near room temperature have more or less internal energy than that? Hint: Their internal energy includes the total energy added in raising the temperature from the boiling point (very low) to room temperature.arrow_forwardAn ideal monatomic gas is taken through the cycle in the PVdiagram. P, kPa - P, kPa where V- 11O, V2= 2.20, P - 98.0 kPa and P= 230 kPa. What is the change in internal energy of the gas as it is taken from A to B?arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning




