
Concept explainers
Interpretation:
It is stated that α-tocopherol is a natural antioxidant that terminates lipid peroxidation by donating H to a lipid radical (
Concept introduction:
In the body, free radicals play important roles in cell signaling and protecting against invading bacteria. Because they are highly reactive, however, these free radicals can lead to unwanted side reactions that damage or kill cells. The hydroxyl radical (
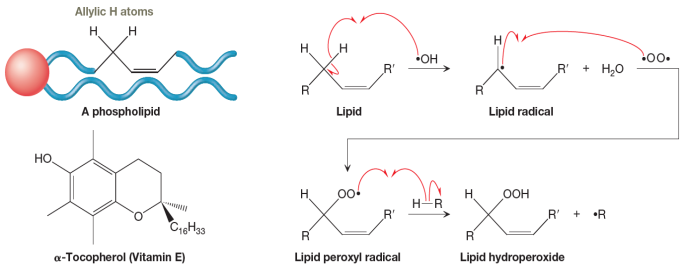
As shown in the scheme above on the right, hydrogen abstraction produces an allylic radical, which subsequently undergoes radical coupling with a molecule of
Lipid peroxides and the compounds they produce in subsequent reactions are very toxic. Fortunately, vitamin E (which is α-tocopherol), found abundantly in foods such as almonds, spinach, and avocados, is a natural antioxidant that is one of the body’s most important defenses against lipid peroxidation. A lipid radical can readily abstract the phenolic
Want to see the full answer?
Check out a sample textbook solution
Chapter 25 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw the next most important resonance structure for the enolate shown. Use curved arrows to show the delocalization of electron pairs in both structures. Include lone pairs of electrons, formal charges, and be sure to draw all hydrogen atoms. You can add condensed hydrogens using the More menu, selecting +H and clicking on the carbon as many times as needed. Draw curved arrows to form the next most important resonance structure. Draw the next most important resonance structure, then draw curved arrows to reform the first structure.arrow_forwardIn the drawing area below, create an acetal with at least 3 methoxy groups, and a total of 5 carbon atoms.arrow_forwardA certain hydrocarbon had the molecular formula C18H30 and contained two triple bonds. Ozonolysis gave CH₂(CH₂)CO₂H and HO₂CCH₂CH₂CO₂H as the only products. Draw a reasonable structure for this hydrocarbon. Click and drag to start drawing a structure.arrow_forward
- I NEED InChI format as the answers !!!!!! 1. For this question you must fill in the structures for the reactive intermediate for the mechanism. First, the overall reaction is shown. Then the steps of the mechanism is shown with letters for the reactive Intermediates. Mechanism arrow are not shown. Draw the structure for the reactive intermediate that forms for the corresponding mechanism step. Use a line structure which means that you should not draw in H atoms and should not enter C for carbons unless necessary. Convert your structures to the InChI format and enter it as your answers.arrow_forwardThe following chemical equation is for an elimination reaction, where the important atoms, groups, and bonds involved have been highlighted for you: O-H + H-0 Use the information provided to predict the missing organic product and draw its structure in the drawing area below. Make sure to use the same skeletal ("line") style for the structure as the rest of the equation.arrow_forwardThe addition of water to aldehydes and ketones occurs rapidly, although it is not thermodynamically favored. What would be the product for the reaction above? Hint: Think of the self-ionization of water and the polarity of the carbonyl group.arrow_forward
- Circle the correct answer in each sentence. Thus sugar is a (simple ; complex) carbohydrate. The linkage marked "a" is a (acetal ; hemiacetal ; acetyl ; hemiacetyl) linkage and is in the (alpha ; beta) configuration. The linkage marked "b" is a (acetal ; hemiacetal ; acetyl ; hemiacetyl) linkage.arrow_forwardPGF2α (Section 4.15) is synthesized in cells using a cyclooxygenase enzyme that catalyzes a multistep radical pathway. Two steps in the pathway are depicted in the accompanying equations. (a) Draw in curved arrows to illustrate how C is converted to D in Step [1]. (b) Identify Y, the product of Step [2], using the curved arrows that are drawn on compound D.arrow_forwardIn the drawing area below, create an acetal with 1 propoxy group, 1 ethoxy group, and a total of 13 carbon atoms.arrow_forward
- Complete the electron‑pushing mechanism for the given base‑promoted hydrolysis of an ester. Add atoms, bonds, nonbonding electron pairs (lone pairs), charges, and curved arrows where indicated.arrow_forwardDraw the structure(s) of the major organic product(s) of the following reaction. H H You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • If no reaction occurs, draw the organic starting material. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate multiple products using the + sign from the drop-down menu. 1. LIAIH4 / dry Et₂0 2. aqueous HCI 2 OOD OO. [F ChemDoodleⓇ < 26arrow_forward4. Given the following condensed structural formulas, a) draw the skeletal structures of each molecule and place partial charges on atoms in the reactive functional group, b) label each as an expected nucleophile or electrophile based on charges, e) determine if the molecule could be described as a Lewis acid (electron pair acceptor) or a Lewis base (electron pair donor). i) NaC=CCH3 ii) C6HsCHO iii) CH3CH₂COOHarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
