
Interpretation:
The product of the reaction for given two reactants should be drawn.
Concept introduction:
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
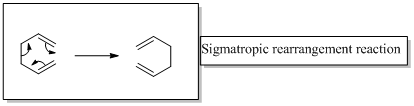
Sigma tropic rearrangement reactions are designated with digits. For example a [1, 3] sigma tropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for sigma tropic rearrangement reactions are listed below
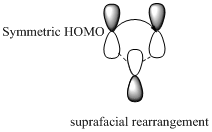
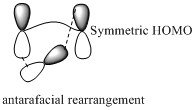
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction. Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Carbon migrating with one lobe of its p orbital interacting

Carbon migrating with both lobe of its p orbital interacting

Trending nowThis is a popular solution!

Chapter 28 Solutions
Organic Chemistry (8th Edition)
- 3. Both El and E2 reactions are possible with 3° alkyl halides. Which elimination mechanism would be favored by the base NaOH? Show the curved arrow mechanism leading to both regioisomers. Draw the energy diagram for this reaction and include the transition state structure leading to each of the possible products. Indicate which one would be favored and use the energy diagram to explain your reasoning. How would you favor the other regioisomer? Br z odd NaOHarrow_forwardFor each section, circle the mechanism from the two options given (E1 or E2) and draw the main organic product resulting from that mechanism. Indicate the stereochemistry and if two configurational isomers form, draw both.arrow_forwardDraw the major product of the following sequence of reactions. If the reaction is likely to give you a mixture of ortho and para disubstituted compounds you should assume that they can be separated; continue the synthesis with either one. If so, draw the intermediates as well. CH3 K₂Cr₂07 Cl₂, AlCl3 H2SO4arrow_forward
- 3. For each reaction, determine all reasonable products that would form. • Underneath each product label if the product is from an Sn1, Sn2, E1, or E2 reaction. In reactions that give both elimination and substitution products, determine whether there will be more elimination or substitution. Br • • For any E1 or Sn1, show any reasonable products of rearrangements. Circle the major product in each reaction If more than one elimination product is formed for a reaction, place a box around the one that is the major elimination product. H I H-C-H CI ax OTS Br Br A Br Na to Хон X DMSO Хон NaOH DMSO OH OHarrow_forwardis this an E1 or E2 mechanism for this reaction? What is the major product and mechanism for it?arrow_forwardIndicate what conditions would be required for each reaction shown below in order for the overall conversion to occur. Draw the intermediate product for each step.arrow_forward
- Rank the following compounds in order of fastest reaction when treated with H2SO4 (1–5, where 1 = fastest), and explain your answer.arrow_forwardThe reaction shown below proceeds by both an SN1 and an SN2 mechanism. Draw the products of each process and use mechanistic arguments to account for any differences in the products formed. Ph 'Br SH Basearrow_forwardIn the box to the left of each reaction below, write the mechanism by which it occurs (could be SN1, SN2, or E1, or even 2 of them). Then draw the product(s).arrow_forward
- Complete the following reactions by providing the missing product(s). Determine what mechanism operates in each case (SN1, SN2, E1, or E2). Show the stereochemistry of the product(s) where appropriate. If more than one product is formed circle the major one.arrow_forwardDraw the products (including stereochemistry) of the following reactions.arrow_forwardDraw the major 1,2-addition product and the major 1,4-addition product of the following reaction. Which product is formed at a high temperature? Which product is the thermodynamic product?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
