
Concept explainers
Interpretation:The correct positioned substituent in the cyclohexane derivative should be chosen.
Concept introduction:In chair form, all the
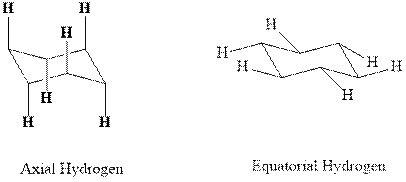
Usually the stable conformations have bulkier groups such as isopropyl,
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: Structure and Function
- 41. Draw the structure of a hydrocarbon that has six carbon atoms and a. three vinylic hydrogens and two allylic hydrogens. b. three vinylic hydrogens and one allylic hydrogen. c. three vinylic hydrogens and no allylic hydrogens.arrow_forwardAdd lone pairs to compound B. В. Select Draw Rings More .C. H3C NH2 // CH Narrow_forwardHere is the chemical structure of methyl propanamide: H H. H H. H-C- C-C -N-C-H H. Decide whether each molecule in the table below is another mnolecule of methyl propanamide, a molecule of an isomer of methyl propanamide, or a molecule of an entirely different compound. molecule relationship to methyl propanamide NH-CH, (Choose one) another molecaule of methyl propanamide a molecule of an isomer of methyl propanamide a molecule of a different compound CH,-CH,-C=0 H H :o: H (Choose one) H-C H- H :0: H H H C-C (Choose one) H. H 回 山回arrow_forward
- The hydrocarbon in which all the 4 valencies of carbon are fully occupied is called as... Select one: O a. Benzene O b. Alkene O C. Alkyne O d. Alkanearrow_forwardConsider a molecule of methanimine (CH3N) compared to a molecule of methylamine (CH5N). The C-N bond in methanimine is than the C-N bond in methylamine. A. longer and stronger B. longer and weaker C. shorter and stronger D. shorter and weaker E. the same strength and length asarrow_forward3. How many valence electron does carbon have? А. 1 В. 2 С. 4 D. 6 4. Which of the following is INCORRECT about the number of bonds one C atom can form? A. 2 double bonds B. 1 triple bond, 3 single bonds C. 2 single bonds, 1 double bond D. 3 single bonds, 1 double bondarrow_forward
- What is the organic product? Cl IV C1 Select one: A. III B. I C. II D. V E. IV :O-CH3 || |||arrow_forwardI. Each -OH group of an alcohol can stabilize four to five carbon atoms.II. The boiling point of alcohol is lower than the boiling point of ethers of comparable mass.A. FIRST statement is INCORRECT, SECOND is CORRECTB. BOTH statements are CORRECTC. FIRST statement is CORRECT, SECOND is INCORRECTD. BOTH statements are INCORRECTarrow_forwardQ2. The following molecule is useful in combinatorial synthesis. H₂N NH₂ SOMe HN- a. What is the name of this molecule? b. What is the purpose using this molecule? c. Describe how this molecule works in combinatorial synthesis. -NH MeOS- HN-arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
