
Concept explainers
Interpretation: The illegal arrows in below set of arrows should be determined.
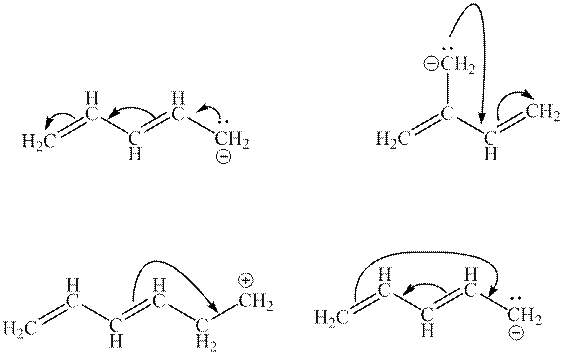
Concept introduction: Lewis structure is a representation of molecules in which dots are shown to represent unshared electrons and lines are shown to represent bonds. These lines and dots represent distribution of electrons in the molecule.
When one single structure is unable to describe all the properties of a single-molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Rules to form resonance structure are as follows:
1. Use arrow types 1 and 2 for the resonance structure of anions in the movement of negative charge.
2. Use only arrow type 3 to move a positive charge for the resonance structure of cations.
3. The sigma bond should not be broken. Any atom must not move from its place and total number of electrons must be the same in all resonance structures.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- (a) Draw an energy diagram for the cyclopropenyl MOs. (The polygon rule is helpful.) Label each MO as bonding, nonbonding, or antibonding, and add the nonbonding line. Notice that it goes through the approximate average of the MOs.(b) Add electrons to your energy diagram to show the configuration of the cyclopropenyl cation and the cyclopropenyl anion. Which is aromatic and which is antiaromatic?arrow_forwardPlease answer all parts to this problem.arrow_forwardAnswer only the 2nd problem. thank you!arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
