
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 24Q
Here are structural formulas for ethane, ethene (ethylene), and ethanol.
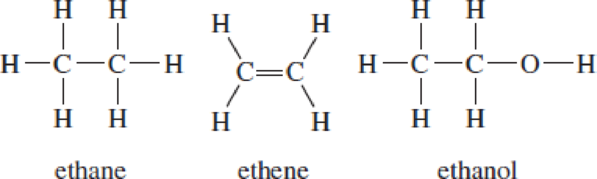
- a. Is ethane an isomer of ethene? Of ethanol? Explain
- b. Are any other isomers possible for ethene? Explain
- c. Are any other isomers possible for ethanol? Explain
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. What is the common name of 2-propanol?
A. Isopropyl alcohol
B. Propyl alcohol
C. sec-propyl alcohol
D. All of the choices
2. Alcohols can be prepared by hydration of alkenes and hydrogenation of aldehydes and ketones. These reactions can be classified as addition reactions.
A. Both statements are correct
B. Both statements are incorrect
C. Only the first statement is correct
D. Only the second statement is correct
3. Which of the following is not an example of an IUPAC name?
A. Methoxycyclohexane
B. Ethyl mercaptan
C. 3-Pentanethiol
D. None of the choices
1. Which of the following fuel has the lowest heat of combustion?a.Methaneb.Octanec.Coald.Ethanol
2. What is the process of breaking larger hydrocarbon molecules into smaller ones at low temperature through the use of catalyst in order to obtain higher quality of gasoline?a.Thermal crackingb.Catalytic crackingc.Catalytic reformingd.Catalytic combination
3. What is the most common oxidizing agent available in the atmosphere that can be used to generate fire?a.hydrogen peroxideb.oxygenc.ozoned.nitrous oxide
1 Which of the following has(have) the same molecular formula as hexane? A. 3-ethylpentane
B. 2,4-dimethylpentane
C. 3,3-dimethylpentane
D. 2,3-dimethylpentane
E. 2,3-dimethylbutane
1b.
Which of these formulas is(are) not theoretically possible? A. C6H6
B. C6H10 C. C6H12 D. C6H14 E. C6H16
1c.
Which of these formulas is(are) not valid for hexane?
A. C6H14
B. C6H6C. C6H16D. CH3CH2CH2CH2CH2CH3
E. CH3(CH2)4CH3
Chapter 5 Solutions
Chemistry In Context
Ch. 5.1 - Prob. 5.1YTCh. 5.1 - Prob. 5.2YTCh. 5.1 - Consult the interactive trends found in the...Ch. 5.1 - Prob. 5.4YTCh. 5.2 - For each of the fuels below, write the balanced...Ch. 5.2 - Prob. 5.6YTCh. 5.3 - Prob. 5.7YTCh. 5.4 - Prob. 5.8YTCh. 5.4 - Prob. 5.9YTCh. 5.4 - Scientific Practices Coal Versus Ethanol On the...
Ch. 5.5 - Prob. 5.12YTCh. 5.5 - Prob. 5.13YTCh. 5.6 - Although power plants require several steps to...Ch. 5.7 - Prob. 5.15YTCh. 5.7 - Prob. 5.16YTCh. 5.7 - An input of energy can be used to decrease entropy...Ch. 5.8 - Prob. 5.18YTCh. 5.8 - Prob. 5.19YTCh. 5.8 - Prob. 5.20YTCh. 5.8 - Prob. 5.21YTCh. 5.10 - Prob. 5.22YTCh. 5.11 - The combustion of one gram of natural gas releases...Ch. 5.11 - a. During the extraction of natural gas, the...Ch. 5.12 - Prob. 5.25YTCh. 5.13 - Prob. 5.26YTCh. 5.13 - Beginning in the 1920s, the octane-booster...Ch. 5.15 - Prob. 5.28YTCh. 5.15 - Prob. 5.29YTCh. 5.16 - Prob. 5.30YTCh. 5.17 - Have you ever been served cherries Jubilee or...Ch. 5.17 - Prob. 5.34YTCh. 5 - Prob. 1QCh. 5 - Prob. 2QCh. 5 - Prob. 3QCh. 5 - Energy exists in different forms in our natural...Ch. 5 - A coal-burning power plant generates electrical...Ch. 5 - Prob. 6QCh. 5 - Prob. 7QCh. 5 - Prob. 8QCh. 5 - Mercury (Hg) is present in trace amounts in coal,...Ch. 5 - Prob. 10QCh. 5 - Here are the condensed structural formulas for two...Ch. 5 - Prob. 12QCh. 5 - Prob. 13QCh. 5 - Consider these three hydrocarbons: At room...Ch. 5 - During petroleum distillation, kerosene and...Ch. 5 - Prob. 16QCh. 5 - a. Write the balanced chemical equation for the...Ch. 5 - Prob. 18QCh. 5 - Prob. 19QCh. 5 - State whether these processes are endothermic or...Ch. 5 - Use the bond energies in Table 5.1 to calculate...Ch. 5 - Use the bond energies in Table 5.1 to calculate...Ch. 5 - Ethanol can be produced by fermentation. Another...Ch. 5 - Here are structural formulas for ethane, ethene...Ch. 5 - These three compounds all have the same chemical...Ch. 5 - Catalysts speed up cracking reactions in oil...Ch. 5 - Explain why cracking is a necessary part of the...Ch. 5 - Consider this equation representing the process of...Ch. 5 - Prob. 29QCh. 5 - Consider these three alcohols: methanol, ethanol,...Ch. 5 - Prob. 31QCh. 5 - Prob. 32QCh. 5 - Prob. 33QCh. 5 - Compare and contrast a molecule of biodiesel with...Ch. 5 - Use Figure 5.6 to compare the energy released for...Ch. 5 - Prob. 36QCh. 5 - The sustainability of burning coal (and other...Ch. 5 - In this chapter, we approximated the chemical...Ch. 5 - Prob. 39QCh. 5 - Compare the processes of combustion and...Ch. 5 - How might you explain the difference between...Ch. 5 - Write a response to this statement: Because of the...Ch. 5 - The concept of entropy and probability is used in...Ch. 5 - Bond energies such as those in Table 5.1 are...Ch. 5 - Use the bond energies in Table 5.1 to explain why...Ch. 5 - Prob. 46QCh. 5 - Prob. 47QCh. 5 - Prob. 48QCh. 5 - Prob. 49QCh. 5 - Prob. 50QCh. 5 - Prob. 51QCh. 5 - Prob. 52QCh. 5 - Prob. 53QCh. 5 - Use a diagram to show the relationship among these...Ch. 5 - On a timescale of a few years, the combustion of...Ch. 5 - Emissions of some pollutants are lower when...Ch. 5 - Although coal contains only trace amounts of...Ch. 5 - Prob. 58QCh. 5 - An article in Scientific American pointed out that...Ch. 5 - C. P. Snow, a noted scientist and author, wrote an...Ch. 5 - Chemical explosions are very exothermic reactions....Ch. 5 - Prob. 63QCh. 5 - Tetraethyllead (TEL) was first approved for use in...Ch. 5 - Tetraethyllead (TEL) has an octane rating of 270....Ch. 5 - Another type of catalyst used in the combustion of...Ch. 5 - Figure 5.8 shows energy differences for the...Ch. 5 - Prob. 68Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
During the early part of the 20th century, sulfanilamide (an antibacterial drug) was only administered by injec...
Elementary Principles of Chemical Processes, Binder Ready Version
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
covered a synthesis of alkynes by a double dehydrohalogenation of dihalides. A student tried to convert trans-2...
Organic Chemistry (9th Edition)
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
Answer: _____
Organic Chemistry As a Second Language: Second Semester Topics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Why are different conformations of an alkane not considered structural isomers?arrow_forwardThe general formula of an alkane is CnH2n+2 . What is the general formula of an (a) alkene? (b) alkyne? (c) alcohol derived from an alkane?arrow_forwardWhich of the following statements is correct? Select one: a.Benzene is an alkene. b.A saturated hydrocarbon would be an alkene. c.An alkene is a hydrocarbon with carbon-carbon double bonds. d.An alkene is a hydrocarbon with carbon-carbon triple bonds.arrow_forward
- 1a. . Isomers are responsible for the diversity of organic compounds. true or false? b. Positional isomers are molecules with different arrangements of the carbon skeleton but with the same chemical formula. true or false? c.)arrow_forwardThe compound CH2 = CH − CH2 − CH2 − CH3 is an example of:a. a pentane.b. a hexene.c. an alkene.d. organic macromolecule.arrow_forwardWhat are structural isomers? What is thesmallest alkane that has a structural isomer?Draw structures to illustrate the isomers.arrow_forward
- 1. Give the complete classification of these organic molecules. If is an amine or alcohol, give the subclass also (primary, secondary, or tertiary). If a hydrocarbon, classify as alkane, alkene, alkyne, or aromatic. a. CH CH CCCH, b. CH,C(OH)(CH)CH₂CH, c. CH CH CH N(CH)CH, 2. Draw the full structure (all atoms and bonds) for the product molecule in each of these organic reactions and give the class or type of each reaction. a. CHỊCH=CHCH(CH)CH, + HO à b. CH,CH(OH)CH₂CH, [O] à c. CHỊCH,CH,COOH+ CHÍNH àarrow_forwardWhat is an alkane? A. A hydrocarbon containing carbons joined only by single bonds B. A hydrocarbon containing a carbon-carbon double bond OC. A hydrocarbon containing an aromatic ring OD. A hydrocarbon containing a carbon-carbon triple bondarrow_forwardWrite structural formulas for all organic compounds. State the type of reaction. Complete and balance the reaction. Name the products. Section 1. propene + hydrogen ? 2. cyclohexane + chlorine ? 3. benzene + iodine ?arrow_forward
- 1. The structures of three compounds are shown in the picture. Why do these substances all belong to the same homologous series? A They all contain an even number of carbon atoms. B They all contain the same functional group. C They are all hydrocarbons. D They are all saturated.arrow_forward1. Using the grignard reaction of alkanes what is the resulting alkane if the reactant is C4H9Br? a. ethane b. propane c. butane d. pentane 2. Using the grignard reaction of alkanes what is the resulting alkane if the reactant is C5H11F? a. ethane b. propane c. butane d. pentane 3. Using Cl2 in C2H4Cl2 will result in HCl and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. not posiblearrow_forward16. Which of the following chemical formulas is best used for isomers such as butane and isobutane?A. Empirical formulaB. Molecular formulaC. Crystal formulaD. Structural formula17. Which of the following scenarios DOES NOT point to the procurement of an assay?A. a certain type of grass is collected from a forest, and was analyzed for its overall mercury (Hg) content in its rootsB. a blood sample was taken from a dead dolphin for a complete genome sequencingC. human saliva is collected for the testing of covid-19 genomeD. a sample of urine was collected from a suspected drunk driver for analysis of phenolic compounds18. Jet is an undergraduate chemistry student, he’s out in the laboratory trying to determine the volatile organic compounds as well as overall protein content of the leaf and stem of a malunggay(Moringa oleifera). He subjected the leaf and stem in a separate digestion reaction (treatment of sulfuric acid), afterwards he subjected the products to high temperature induction…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License