
Concept explainers
Interpretation:
The
Concept Introduction:
Esters are prepared by condensation of
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
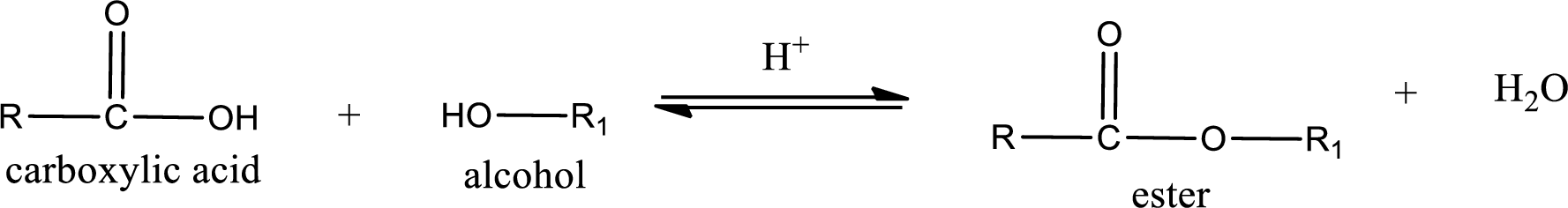
In the above reaction, hydroxyl group is lost from carboxylic acid and hydrogen is lost from alcohol molecule to form water as the byproduct.
If the considered compound is a hydroxyacid, then there is a possibility of intermolecular esterification reaction. The product obtained in this case is a cyclic ester. Cyclic esters contain an oxygen atom in the ring and a carbonyl group also.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic And Biological Chemistry
- Would you expect a molecule of urea produced in the body to have any different physical or chemical properties from a molecule of urea prepared in a laboratory?arrow_forwardFats belong to the class of organic compounds represented by the general formula, RCOOR', where R and R' represent hydrocarbon groups; therefore, fats are: a. ethers. b. soaps. c. esters. d. lipases.arrow_forwardFats belong to the class of organic compounds represented by the general formula, RCOOR', where R and R' represent hydrocarbon groups. What is the name of the functional group present in fats? What functional group is common to all saponifiable lipids?arrow_forward
- 4. CAN CARBOXYLIC ACIDS BE FURTHER OXIDIZED? WHY OR WHY NOT? 5. HOW IS SOAP PREPARED? 6. WHY IS PREPARATION OF AN ESTER REFFERED TO AS A CONDENSATION REACTION?arrow_forwardWhat is produced from triacylglycerol that forms a mixture of long chain carboxylate salts?arrow_forwardthe chemical formula for sodium thiosulfate pentahydrate.arrow_forward
- Give an example of (a) polyester (b) polyamide.arrow_forwardWhat is the product of the ionization reaction of butanoic acid (CH3CH2CH2COOH) in water? Including all hydrogen atoms and charges.arrow_forwardWhat is the structural formula of glycerol? To which organic function do these molecules belong?arrow_forward
- What are the structures of cyclohexanone and salicylaldehyde substances constitutes? i) Is it saturated or unsaturated or aromatic amine or phenol? ii) Aromatic or Aliphatic in nature? iii) What is their functional group? (aldehyde/ketones/esters)arrow_forwarda)why can esterification also be considered to be a neutralization reaction? b)What is distillation? How would distillation be used to purify an ester?arrow_forwardwhat makes acetone,Ethanol,and Sucrose compounds soluble in water?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





