
(a)
Interpretation:
The bond among pair of compounds having higher
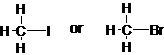
Concept Introduction:
The bond dissociation energy is defined as the enthalpy change occurs when a covalent bond is broken by equally dividing the electrons between the two atoms in the bond. The stronger the bond, the higher is its dissociation energy.
(b)
Interpretation:
The bond among pair of compounds having the higher bond dissociation energy needs to be determined.
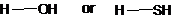
Concept Introduction:
The bond dissociation energy is defined as the enthalpy change occurs when a covalent bond is broken by equally dividing the electrons between the two atoms in the bond. The stronger the bond, the higher is its dissociation energy.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Choose the more stable, i.e., less reactive, molecule (or ion) in each of the three pairs of structures drawn below: H2N NH2 EN-H но 1. 3. 4. 6. O 1, 3 & 6 O 2, 3 & 5 1, 4 & 5 2, 3 & 6 2, 4 & 5 oving to another question will save this response. 5. 2.arrow_forwardProblem 10(2.48) Draw all significant resonance structures for each of the following compounds. Problem 10 (=2.48) Draw all significant resonance structures for each of the following compounds. (a) OH H (b) (c)arrow_forward(1, 7) Draw both resonance structures of the anion formed by the reaction of the most acidic C-H bond of the compound below with base. Include all valence lone pairs in your answer. For structures having different hydrogens of comparable acidity, assume that the reaction occurs at the less-substituted carbon. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate resonance structures using the ↔ symbol from the drop-down menu.arrow_forward
- 2.10 Drawing Resonance Structures via Pattern Recognition Which of the following shows correct arrow placement that represents significant resonance for the given species? B 00 H-N- -C-H O H-NH H O None of the choices given represent significant resonance. Ö: H H-NIC-H Save for Later 0=0 H H-NIC-H :0-0 Submit Answerarrow_forward4.1 only asaparrow_forward2. Below is adenine, one of the four nucleobases which make up the alphabet for DNA and RNA. Is the bicyclic system of adenine aromatic? Circle all the pairs of electrons which are basic and draw a box around the pairs of electrons which could be in the pi system. (7 pts) NH2 Narrow_forward
- Problem 3.13 illing point. Which compound in each pair has the higher boiling point? a. b. or OH or More Practice: Try Problems 3.43, 3.44. O H C. d. or Ja CI obo or Problem 3.14 Explain why the boiling point of propanamide, CH3CH₂CONH2, is considerably higher than the boiling point of N,N-dimethylformamide, HCON(CH3)2 (213 °C vs. 153 °C), even though both compounds are isomeric amides.arrow_forward6. (3 points) Give IUPAC names for the following compounds: CH3 с сarrow_forward6.1.3 Draw the product(s) of the dehydration reaction H- OH -H20 H- HO CH;OHarrow_forward
- ____GaF3 + ___Cs--> ____CsF + ____Ga A. (2,2,3,1) B. (1,3,2,1) C. (1,3,3,1) D. (2,3,1,3) E. (1,2,3,1)arrow_forward(2.45 What is the Ka for each compound? Use a calculator when necessary. а. H2S b. CICH,COOH C. HCN pk, = 7.0 pka = 2.8 pka 9.1arrow_forwardSpecify the formal charge at each of the labeled atoms, a - c, in each of the following structures. 1. 2. H3C-N=N=N: a b c H. H I H-C II H H d_d H C H-C H H a Ze H C-H H H C c H -H H a. a. b. b. C. 9arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
