
Concept explainers
Write a Lewis formula for each of the following organic molecules:
(vinyl chloride: starting material for the preparation of PVC plastics)
(halothane: a nonflammable inhalation anesthetic; all three fluorines are
bonded to the same carbon)
(Freon 114: formerly used as a refrigerant and as an aerosol propellant; each
carbon bears one chlorine)
Interpretation:
The Lewis formula for each of the given organic molecules is to be written.
Concept introduction:
When writing the Lewis formula for a molecule, the total number of valence electrons in the molecule is first determined based on its molecular formula.
The bonded atoms are connected by a shared pair of electrons which is shown by a dash.
The number of electrons in the bonds is subtracted from the total number of valence electrons. This gives the number of electrons that remain to be added in the formula.
These remaining electrons are added as unshared electron pairs in such a way that the most electronegative atoms have eight electrons around them.
Second-row elements cannot have more than eight valence electrons.
Answer to Problem 41P
Solution:
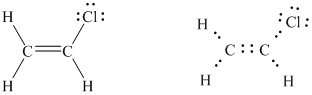
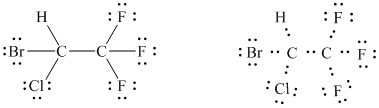
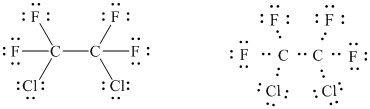
Explanation of Solution
a)
The total number of valence electrons in
The basic framework of the given molecule can be drawn as follows.
The above figure shows the presence of
Three pairs of unshared electrons are assigned to the chlorine atom and one pair of unshared electrons is assigned on one of the carbon atoms as follows.
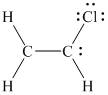
The octet of each atom in the molecule, except one carbon atom, is complete. Hence, to complete the octet of this one carbon atom, the unshared pair of electrons on the other carbon atom shifts towards the bond between the two carbon atoms resulting in the formation of a double bond.
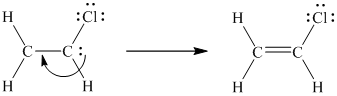
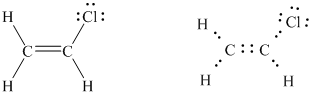
Now the octet of each atom is complete. In this way, the Lewis structure of vinyl chloride was written.
b)
bonded to the same carbon)
The basic framework can therefore, be drawn as follows.
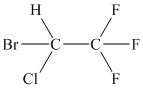
It is evident from the figure drawn for the given molecule that there are a total of
Of these
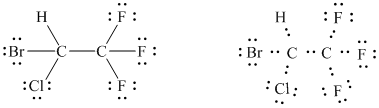
In all,
In this way, the Lewis structure of
c)
carbon bears one chlorine)
The basic framework of the molecule can be drawn as follows.
The above figure shows that there are
Three pairs of unshared electrons are assigned on each halogen atom in order to complete their octets.
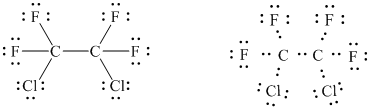
In all,
In this way, the Lewis structure of
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry - Standalone book
Additional Science Textbook Solutions
Essential Organic Chemistry (3rd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
Chemistry
EBK INTRODUCTION TO CHEMISTRY
- An ionic compound has the following composition (by mass): Mg, 10.9%; Cl, 31.8%; O, 57.3%. What are the formula and name of the compound? Write the Lewis formulas for the ions.arrow_forwardAn ionic compound has the following composition (by mass): Ca, 30.3%; N, 21.2%; O, 48.5%. What are the formula and name of the compound? Write the Lewis formulas for the ions.arrow_forwardA liquid compound used in dry cleaning contains 14.5% C and 85.5% Cl by mass and has a molecular mass of 166 amu. Write the Lewis formula for the molecule.arrow_forward
- Draw Lewis structures for the following molecules or polyatomic ions. a. SO3 b. SO32arrow_forwardDefine the term lattice energy. Why, energetically, do ionic compounds form? Fig. 3-8 illustrates the energy changes involved in the formation of MgO(s) and NaF(s). Why is the lattice energy of MgO(s) so different from that of NaF(s)? The magnesium oxide is composed of Mg2+ and O2 ions. Energetically, why does Mg2+O2 form and not Mg+O? Why doesnt Mg3+O3 form?arrow_forwardEach compound contains both ions and covalent bonds. Draw the Lewis structure for each compound. Show with dashes which are covalent bonds and show with charges which are ions. (a) Sodium methoxide, CH3ONa (b) Ammonium chloride, NH4Cl (c) Sodium bicarbonate, NaHCO3 (d) Sodium borohydride, NaBH4 (e) Lithium aluminum hydride, LiAlH4arrow_forward
- Four hypothetical elements, A, B, C, and D, have electronegativities A = 3.8, B = 3.3, C = 2.8, and D = 1.3. These elements form the compounds BA, DA, DB, and CA. Arrange the compounds in order of a. Increasing ionic bond character b. Decreasing covalent bond characterarrow_forwardDraw Lewis symbols for the following ions. a. O2 b. S2 c. Si4 d. Clarrow_forwardA polyatomic ion is composed of C, N, and an unknown element X. The skeletal Lewis structure of this polyatomic ion is [XCN]. The ion X2 has an electron configuration of [Ar]4s23d104p6. What is element X? Knowing the identity of X, complete the Lewis structure of the polyatomic ion, including all important resonance structures.arrow_forward
- Consider the pyrosulfate ion, S2O72-. It has no sulfur–sulfur nor oxygen–oxygen bonds. (a) Write a Lewis structure for the pyrosulfate ion using only single bonds. (b) What is the formal charge on the sulfur atoms for the Lewis structure you drew in part (a)? (c) Write another Lewis structure using six bonds and two O—S bonds. (d) What is the formal charge on each atom for the structure you drew in part (c)?arrow_forwardBorazine (B3N3H6) has often been called inorganic benzene. Write Lewis structures for borazine. Borazine contains a six-membered ring of alternating boron and nitrogen atoms with one hydrogen bonded to each boron and nitrogen.arrow_forwardNitrosyl azide, N4O, is a pale yellow solid first synthesized in 1993. Write the Lewis structure for nitrosyl azide.arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





