
(a)
Interpretation:
The synthesis of
Concept introduction:
Oxymercuration reaction is a type of reaction in which an
Answer to Problem 11.61AP
The synthesis of
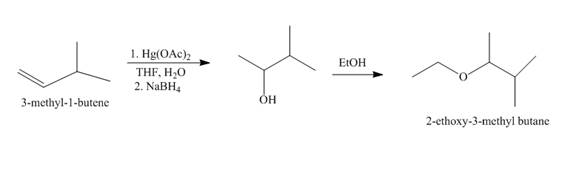
Explanation of Solution
The mercuric acetate

Figure 1
The route of synthesis of the formation of
(b)
Interpretation:
The synthesis of
Concept introduction:
The substances which on addition removes oxygen atom or hydrogen atom from the other substance, that is, reduces the other substances are known as reducing agents. Reducing agents themselves get oxidized. Strong reducing agents like
Answer to Problem 11.61AP
The synthesis of
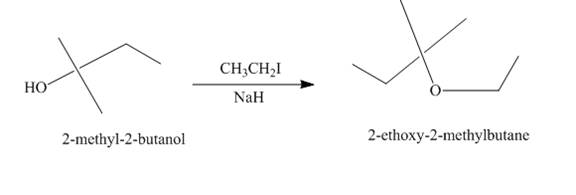
Explanation of Solution
The treatment of
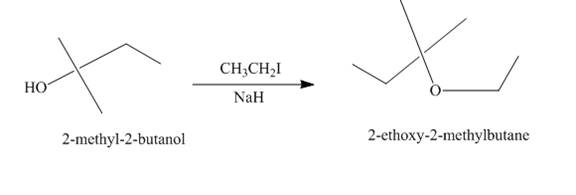
Figure 2
The route of synthesis of the formation of
(c)
Interpretation:
The synthesis of
Concept introduction:
Grignard reagents are
Answer to Problem 11.61AP
The synthesis of
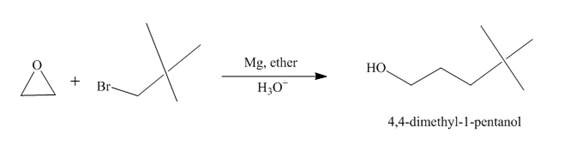
Explanation of Solution
The reaction of tert-butyl bromide with

Figure 3
The route of synthesis of the formation of
(d)
Interpretation:
The synthesis of the given compound from the compounds that contain
Concept introduction:
Oxidation is the process in which there is an addition of oxygen atom or removal of hydrogen atom. The agents used for the oxidation reaction is known as oxidizing agents. It oxidizes the other substance and gets reduced in the reaction. The common oxidizing agents are nitric acid, oxygen and potassium dichromate.
Answer to Problem 11.61AP
The synthesis of the given compound from the compounds that contain
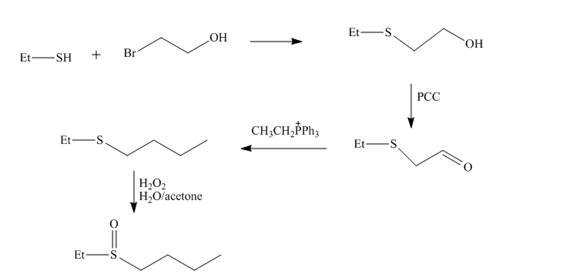
Explanation of Solution
The reaction of ethnethiol with
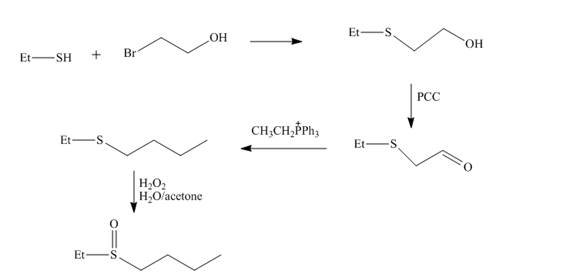
Figure 4
The synthesis of the given compound from the compounds that contain
(e)
Interpretation:
The synthesis of the given compound from an alkene is to be drawn.
Concept introduction:
Oxidation is the process in which there is an addition of oxygen atom or removal of hydrogen atom. The agents used for the oxidation reaction is known as oxidizing agents. It oxidizes the other substance and gets reduced in the reaction. The common oxidizing agents are nitric acid, oxygen and potassium dichromate.
Answer to Problem 11.61AP
The synthesis of the given compound from an alkene is shown below.
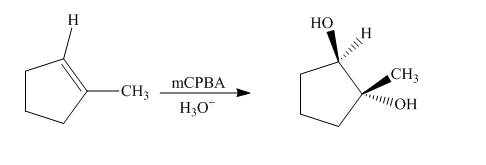
Explanation of Solution
The reaction of

Figure 5
The synthesis of the given compound from an alkene is shown in Figure 5.
(f)
Interpretation:
The synthesis of cyclohexyl isopropyl ether from cyclohexene is to be drawn.
Concept introduction:
Oxidation is the process in which there is an addition of oxygen atom or removal of hydrogen atom. The agents used for the oxidation reaction is known as oxidizing agents. It oxidizes the other substance and gets reduced in the reaction. The common oxidizing agents are nitric acid, oxygen and potassium dichromate.
Answer to Problem 11.61AP
The synthesis of cyclohexyl isopropyl ether from cyclohexene is shown below.
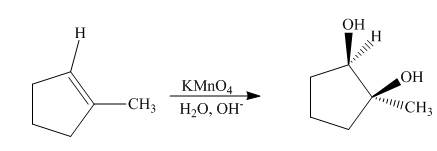
Explanation of Solution
The compound,

Figure 6
The synthesis of cyclohexyl isopropyl ether from cyclohexene is shown Figure 6.
(g)
Interpretation:
The synthesis of cyclohexyl isopropyl ether from cyclohexene is to be drawn.
Concept introduction:
Oxymercuration reaction is a type of reaction in which an alkene gets converted to alcohol. The mercuric acetate is used in the reaction as a reagent. This reagent attacks the alkene to form a cyclic intermediate compound which further undergoes reduction to form alcohol.
Answer to Problem 11.61AP
The synthesis of cyclohexyl isopropyl ether from cyclohexene is shown below.
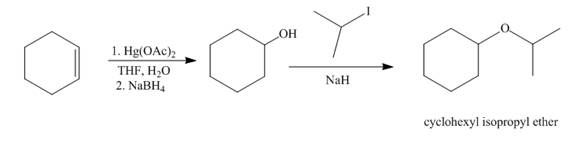
Explanation of Solution
The mercuric acetate

Figure 7
The synthesis route of cyclohexyl isopropyl ether from cyclohexene is shown in Figure 7.
(h)
Interpretation:
The synthesis of the given compound from
Concept introduction:
Grignard reagents are organometallic compounds which are prepared using alkyl halides in the presence of magnesium metal in dry ether. These reagents act as strong nucleophiles and bases
Answer to Problem 11.61AP
The synthesis of the given compound from
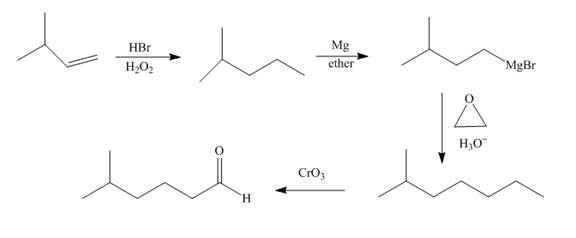
Explanation of Solution
Bromination of alkene occurs with
Grigmard reagent reacts with the ethylene oxide followed by the hydrolysis to form an alcohol which upon oxidation by
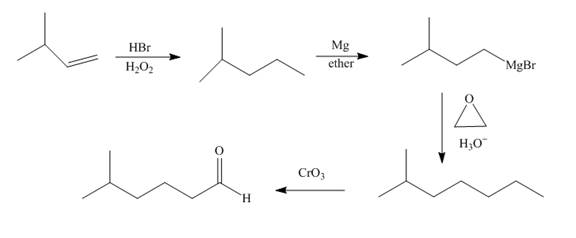
Figure 8
The synthesis of the given compound from
(i)
Interpretation:
The synthesis of the given compound from
Concept introduction:
An
Oxidation is the process in which there is an addition of oxygen atom or removal of hydrogen atom. The agents used for the oxidation reaction is known as oxidizing agents. It oxidizes the other substance and gets reduced in the reaction. The common oxidizing agents are nitric acid, oxygen and potassium dichromate.
Answer to Problem 11.61AP
The synthesis of the given compound from
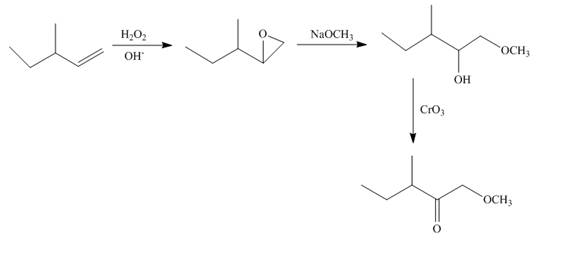
Explanation of Solution
Epoxidation of

Figure 9
The synthesis route of the given compound from
(j)
Interpretation:
The synthesis of the given compound from
Concept introduction:
An epoxide is cyclic ether. It can be prepare by the reaction of alkene with percarboxylic acid with removal of carboxylic acid. An epoxide undergoes ring opening reactions with an acid to give a product with inversion configuration.
Oxidation is the process in which there is an addition of oxygen atom or removal of hydrogen atom. The agents used for the oxidation reaction is known as oxidizing agents. It oxidizes the other substance and get reduced in the reaction. The common oxidizing agents are nitric acid, oxygen and potassium dichromate.
Answer to Problem 11.61AP
The synthesis of the given compound from
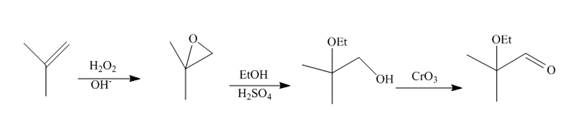
Explanation of Solution
Epoxidation of

Figure 10
The synthesis route of the given compound from
(k)
Interpretation:
The synthesis of given compound from the allyl chloride is to be drawn.
Concept introduction:
The nucleophilic substitution reactions depend upon the nucleophilicity and concentration of the nucleophile. There are two types of nucleophilic substitution reaction.
The
Answer to Problem 11.61AP
The synthesis of given compound from the allyl chloride is shown below.
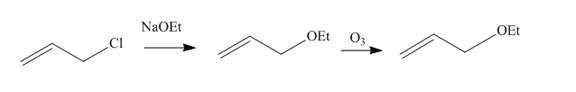
Explanation of Solution
Allyl chloride reacts with

Figure 11
The synthesis of given compound from the allyl chloride is shown in Figure 11.
(l)
Interpretation:
The synthesis of the given compound is to be drawn.
Concept introduction:
Wolff Kishner Reduction is a reaction in which aldehydes and
Answer to Problem 11.61AP
The synthesis of the given compound is shown below.
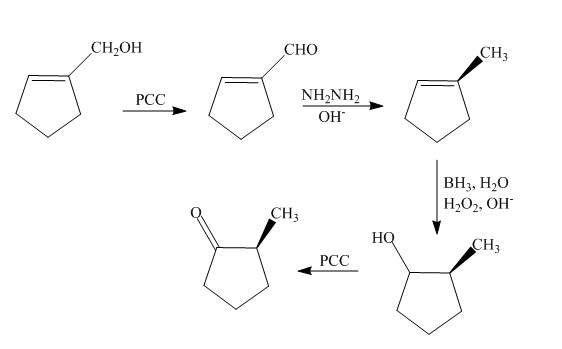
Explanation of Solution
The reaction of cyclopentenyl methanol with

Figure 12
The synthesis of the given compound is shown in Figure 12.
(m)
Interpretation:
The synthesis of the given compound is to be drawn.
Concept introduction:
The substances which on addition removes oxygen atom or hydrogen atom from the other substance, that is, reduces the other substances are known as reducing agents. Reducing agents themselves get oxidized. Strong reducing agents like
Answer to Problem 11.61AP
The synthesis of the given compound is shown below.
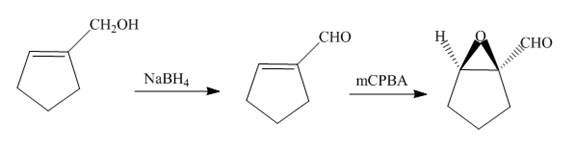
Explanation of Solution
The alcohol is reduced in presence of

Figure 13
The synthesis of the given compound is shown in Figure 13.
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry
- Show how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures.(a) ethoxybenzene (b) 1,2-dichloro-4-nitrobenzene (c) 1-phenylpropan-2-olarrow_forwardDisiamylborane adds only once to alkynes by virtue of its two bulky secondary isoamylgroups. Disiamylborane is prepared by the reaction of BH3 # THF with an alkene.(a) Draw the structural formulas of the reagents and the products in the preparation ofdisiamylborane.(b) Explain why the reaction in part (a) goes only as far as the dialkylborane. Why isSia3B not formed?arrow_forward(b) acid, HNO3 to produce compound L. The reaction of compound L with bromine, Br2 in the presence of iron tribromide, FeBr3 produced compound M. Benzene also undergoes Fridel-crafts alkylation reaction with chloroethane, CH;CH2CI using catalyst N to produce compound P. Benzene, CeHe undergoes substitution reaction with concentrated nitric Benzena, CsHe menjalani tindak balas penukargantian dengan asid nitrik, HNO, pekat untuk menghasilkan sebatian L. Tindak balas sebatian L dengan bromin, Brz dengan kehadiran ferum tribromida, FeBrs menghasilkan sebatian M. Benzena juga menjalani tindak balas alkilasi Fridel-crafts dengan kloroetana, CH3CH2CI dengan menggunakan pemangkin N untuk menghasilkan sebatian P. (i) Draw the structural formula of L, M and P. Lukiskan formula struktur bagi sebatian L, M dan P. (ii) State catalyst N. Nyatakan pemangkin N. (iii) Show the formation of electrophile that will be reacted with benzene for the formation of compound P.arrow_forward
- Synthesize from benzene. (Hint: All of these require diazonium ions.)(a) 3-ethylbenzoic acidarrow_forwardConsider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in electrophilic aromatic substitution? (b) Which ring is the least reactive in electrophilic aromatic substitution?arrow_forwardAnswer ALL parts of this question. (a) If ethyne is treated according to the following scheme, the final product is trans-pent-2-ene, as shown: H- (c) C-H (i) NaNH, / NH, (ii) CH₂Br (iii) NaNH, / NH (iv) CH₂CH₂Br (v) Na/NH3 H₂C H H CH₂CH3 Show the structures of the intermediates formed after each of the first four steps and identify the mechanism involved in steps (ii) and (iv). (b) If cis-pent-2-ene was wanted as the final product, how would step (v) in the sequence above must be modified? | The alkene shown in part (a) of this question could also be made by treating 2-bromopentane with base. It would be the major product out of three possible alkenes that could form. Give the structures of the two other alkenes that would form and comment on why trans-pent-2-ene is the major one that forms here.arrow_forward
- Rank the compounds in each group according to their reactivity towardelectrophilic substitution.(a) Chlorobenzene, o-dichlorobenzene, benzene(b) p-Bromonitrobenzene , nitrobenzene, phenol(c) Fluorobenzene, benzaldehyde, a-xylene(d) Benzonitrile, p-methylbenzonitr ile,p-methoxybenzonitrilearrow_forwardShow how you can synthesize the following compounds starting with benzene, toluene, and alcohols containing no morethan four carbon atoms as your organic starting materials. Assume that para is the major product (and separable fromortho) in ortho, para mixtures.(a) pentan-1-amine (b) N-methylbutan-1-amine(c) N-ethyl-N-propylbutan-2-amine (d) N-benzylpropan-1-amine(e)N N OH(e) (f) 3-propylaniline(g) 4-isobutylanilinearrow_forward(a) Why are alkyl halides insoluble in water? (b) Why is Butan-l-ol optically inactive but Butan-2-ol is optically active? (c) Although chlorine is an electron withdrawing group, yet it is ortho-, Para- directing in electrophilic aromatic substitution reaction. Why?arrow_forward
- Starting exactly with any acid chloride with exactly with 5 carbon atoms, and using appropriate reagents outline the synthesis of the following molecules: (a) 2,6-dimethyl-4-heptanone (b) 4-propyl-4-octanolarrow_forward3. (a) Identify the reagents (A-D) in each of the following reactions. OTs OH нос enantiomer HO. (b) The following reaction did not afford compound X. () Explain your answer. (1) Draw the structure of the actual product formed. (c) Device a synthesis for the following product from the given starting material. More than one step is required, CH,Br (d) Propose a stepwise mechanism for the following reaction. но. HBarrow_forwardGuiding your reasoning by retrosynthetic analysis, show how you could prepare each of the following compounds from the given starting material and any necessary organic or inorganic reagents. All require more than one synthetic step. (a) Cyclopentyl iodide from cyclopentane (b) 1-Bromo-2-methylpropane from 2-bromo-2-methylpropane (c) meso-2,3-Dibromobutane from 2-butyne (d) 1-Heptene from 1-bromopentane (e) cis-2-Hexene from 1,2-dibromopentane (f) Butyl methyl ether (CH3CH2CH2CH2OCH3) from 1-butenearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





