
Concept explainers
Interpretation:
The density of a hypothetical ionic compound for given radii and masses of cations and anions, is to be determined.
Concept introduction:
Ionic compounds contain cations and anions that are arranged in a crystal lattice.
A crystal lattice is made of small repeating unit cells.
A unit cell is of primitive or centered type.
Each atom or ion in a unit cell is shared by the adjacent cells. An atom or ions at corners is shared by eight unit cells.
The edge length of a body-centered cubic cell is given by the relation as follows:
Here,
is the edge length,
is the radius of cation, and
The volume of the unit cell
The density
Here,
is the mass.
Answer to Problem 146AP
Solution:
Explanation of Solution
Given information:
Radii of the anion
The figure is as shown below:
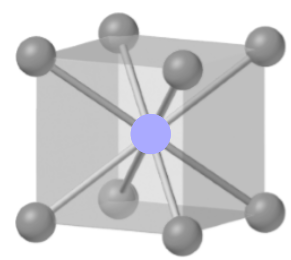
The given unit cell is body-centered, in which there is one cation at the center, and eight anions at the corners, shared by eight unit cells.
The contribution of 8 anions at the corners to one unit cell is:
In the unit cell, one anion and one cation are present.
The mass of an anion is
and that of a cation is
The relation between
and
is as:
Convert the mass to grams as follows:
Similarly,
Thus, the mass of the unit cell will be:
The formula to calculate the edge length is as:
Substitute
for
and
for
in the above expression as:
Convert the edge length to
as follows:
The expression to calculate volume is as:
Substitute
in the above expression as:
Now, finally calculate density as:
Substitute
for
for
in the above expression as:
The density of the hypothetical ionic compound is
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry
- The unit cell of silicon carbide, SiC, is illustrated below. (a) In what type of unit cell are the (dark gray) C atoms arranged? (b) If one edge of the silicon carbide unit cell is 436.0 pm, what is the calculated density of this compound? A portion of the solid-state structure of silicon carbide.arrow_forwardThe CsCl structure is a simple cubic array of chloride ions with a cesium ion at the center of each cubic array (see Exercise 69). Given that the density of cesium chloride is 3.97 g/cm3, and assuming that the chloride and cesium ions touch along the body diagonal of the cubic unit cell, calculate the distance between the centers of adjacent Cs+ and Cl ions in the solid. Compare this value with the expected distance based on the sizes of the ions. The ionic radius of Cs+ is 169 pm, and the ionic radius of Cl is 181 pm.arrow_forwardA portion of the crystalline lattice for potassium is illustrated below. (a) In what type of unit cell are the K atoms arranged? A portion of the solid-state structure of potassium. (b) If one edge of the potassium unit cell is 533 pm, what is the density of potassium?arrow_forward
- Rutile, TiO2, crystallizes in a structure characteristic of many other ionic compounds How many formula units of TiO2 are in the unit cell illustrated here? (The oxide ions marked by an x are wholly within the cell; the others are in the cell faces.) Unit cell for rufflearrow_forwardPotassium chloride has the same unit cell as NaCl. Using the ion sizes in Figure 7.11, calculate the density of KCl. FIGURE 7.11 Relative sizes of some common ions. Rodii are given in picometers (1 pm 1 1012 m). (Data taken from J. Emsley, The Elements, Clarendon Press, Oxford, 1998, 3rd edition.)arrow_forwardCalcium oxide consists of a face-centered cubic array of O2 ions, with Ca2+ ions at the center of the unit cell and along the centers of all 12 edges. Calculate the number of each ion in the unit cell.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





