
Why do ethanol and dimethyl ether have such different properties even though they have same chemical formula?
To determine: Why do ethanol and dimethyl ether have such different properties even when they have same chemical formula?
Answer to Problem 1E
Solution:
The difference is caused because of different functional group present in both the compounds. Ethanol contains alcohol (-OH) functional group while dimethyl ether contains ether (-O-) group.
Explanation of Solution
The physical and chemical properties of any compound is closely related with its structure and bonding pattern.
So, to understand the difference between properties of the given compounds their structural formula are very important to be discussed.
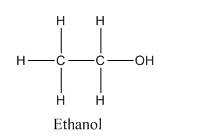
Chemical formula of Ethanol : C2H6O or , C2H5OH
Structural formula of Ethanol :
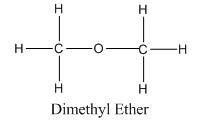
Chemical formula of Dimethyl ether : C2H6O or , CH3-O-CH3
Structural formula of Ethanol :
The difference can be easily seen, that in one hand, where ethanol is a polar molecule with terminal −OH group having electronegativity difference and the ethanol molecules are attached together by hydrogen bonding as the H-atom is directly attached to electronegative O-atom.
While in other hand, dimethyl ether has two methyl groups attached to one other by (-O-) group on either sides which generates a regular dipole-dipole interactions.
The two types of bonding pattern bring changes in the properties of the two compounds.
Also, there are many more functional aspects of both the functional groups which change the properties of compounds instead of having same chemical formula.
The difference in functional groups of ethanol and dimethyl ether brings the difference in their properties.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry: Structure and Properties (2nd Edition)
- hat does temperature measure? Are the molecules in a beaker of warm water moving at the same speed as the molecules in a beaker of cold water? Explain? What is heat? Is heat the same as temperature?arrow_forwardWhat is the enthalpy of formation of any structure in the figure, or any structure that contains the same group?arrow_forward10.) The structural formula of a certain aldehyde (related to formaldehyde) is H3C-CH2-CHO. Draw a Lewis structure for this aldehyde and determine the number of bonds present. Note that a single or a double or a triple bond counts as one bond. Write the number, not the word.arrow_forward
- 5. You are given a list of binary compounds (only two different elements) and asked to determine the relative melting points. a) Which would have the higher melting point, the binary ionic compounds or the binary covalent compounds? Explain your choice? b) If you have two different ionic compounds, like Bal; and Mgo, which would have the higher melting point and why? c) For those compounds that are covalent, which would you expect to have the higher melting point and why (what would you be looking for – give an example)?arrow_forwardhat is meant by the term driving forces? Why are mailer spread and energy spread considered to be driving forces?arrow_forwardWhat is a driving force? Name two common and important driving forces, and give an example of each. What is entropy? Although the total energy of the universe is constant, is the entropy of the universe constant? What is a spontaneous process?arrow_forward
- The structural formula for the open-chain form of glucose is Glucose dissolves readily in water. Use molecular structure principles to explain why glucose is so water-soluble.arrow_forwardCCl4 is a nonpolar molecule, while CHCl3 and CH2Cl2 are polar molecules. Draw the Lewis structures of these three molecules. Explain the observation in polarity of the molecules.arrow_forwardDraw the Lewis structure for hydrogen peroxide, H2O2. Based on this structure, how many polar bonds and non-polar bonds are present?arrow_forward
- A hydrated salt is one in which water molecules are suspended in the crystal lattice structure of the salt. Upon heating what happens to these water molecules? What change can be expected in the structure and appearance of the salt?arrow_forwardwhy is the ability of water to undergo phase charges is significant?arrow_forwardCalculate the heat of sublimation of PI3[PI3(s)→PI3(g)]. The standard heat of formation of PI3(s) is -24.7 kJ/mol and the PI bond energy in this molecule is 184 kJ/mol. The standard heat of formation of P(g) is 334 kJ/mol and that of I2(g) is 62 kJ/mol. The I2 bond energy is 151 kJ/mol.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



