
Concept explainers
Give the IUPAC name for each compound.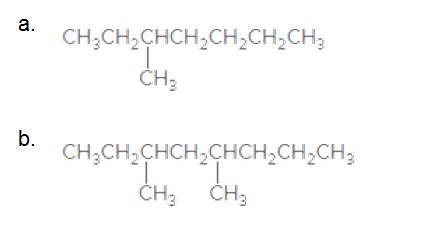
c.
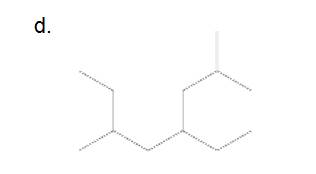
e.
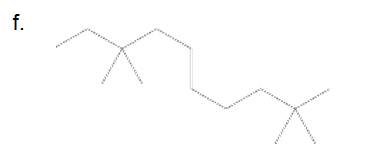
(a)
Interpretation:
The IUPAC nomenclature of alkanes having only one branched-chain needs to be determined.
Concept Introduction:
Every organic compound has its own unique name which is followed by the IUPAC (International Union of Pure and Applied Chemistry). For alkane also, there is a particular rule for IUPAC nomenclature. The rules are −
- The name has to end with suffix "−ane" for all alkanes.
- The longest continuous carbon chain that contains the functional group will be treated as the main chain.
- Number the carbons in the longest carbon chain. As per as the no of carbon present in the longest chain, "Pent (for 5)", "Hept (for 6)" this word will be added before "ane".
- The branched groups present in the longest carbon chain beside the main chain should be named by the number of carbon atoms present in the branched group. These groups will end with "-yl" (e.g.- ethyl, methyl) at their end.
- The position of the group on the main carbon chain and it should be mentioned (e.g. 2,3 -).
- Numbering of the main chain should start from the side having smallest distance with most priority brunch (priority depends on alphabetical order of the brunch).
- The brunched groups should be listed before the name of the main carbon chain in alphabetical order (ignoring prefix like di/tri).
- Combine the elements of the name into a single word in the following order:
- Branched groups in alphabetical order (ignoring prefixes).
- Prefix of main chain
- End with "-ane" suffix.
Answer to Problem 45P
3-Methylheptane
Explanation of Solution
The longest carbon chain has 7 carbon.
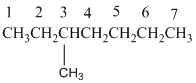
And there is a methyl group in the no 3 carbon. As the branch group has only one CH3, it is called 'Methyl'.
So, following by the above mention rule, the name of the alkane is 3 (the position of the branch) + Methyl (the name of the branch)+hept ( 7 carbon in main chain)+ "-ane"= 3-Methylheptane
(b)
Interpretation:
The IUPAC nomenclature of alkanes having 2 same brunches needs to be determined.
Concept Introduction:
Every organic compound has its own unique name which is followed by the IUPAC (International Union of Pure and Applied Chemistry). For alkane also, there is a particular rule for IUPAC nomenclature. The rules are −
- The name has to end with suffix "−ane" for all alkanes.
- The longest continuous carbon chain that contains the functional group will be treated as the main chain.
- Number the carbons in the longest carbon chain. As per as the no of carbon present in the longest chain, "Pent (for 5)", "Hept (for 6)" this word will be added before "ane".
- The branched groups present in the longest carbon chain beside the main chain should be named by the number of carbon atoms present in the branched group. These groups will end with "-yl" (e.g.- ethyl, methyl) at their end.
- The position of the group on the main carbon chain and it should be mentioned (e.g. 2,3 -).
- Numbering of the main chain should start from the side having smallest distance with most priority brunch (priority depends on alphabetical order of the brunch).
- The brunched groups should be listed before the name of the main carbon chain in alphabetical order (ignoring prefix like di/tri).
- Combine the elements of the name into a single word in the following order:
- Branched groups in alphabetical order (ignoring prefixes).
- Prefix of main chain
- End with "-ane" suffix.
Answer to Problem 45P
3,5-dimethyloctane
Explanation of Solution
The longest carbon chain has 8 carbon.
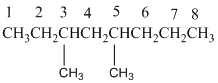
And there are two methyl groups in the no. 3 and no. 5 carbons. As the branch group has only one CH3, it is called 'Methyl'.
So, following by the above mention rule, the name of the alkane is 3,5 (the position of the branch) + dimethyl (the name of the branch, 'di' as two methyl are present)+ oct( 8 carbon in main chain)+ "-ane"= 3,5-dimethyloctane.
(c)
Interpretation:
The IUPAC nomenclature of Alkanes having 2 same brunches in same carbon needs to be determined.
Concept Introduction:
Every organic compound has its own unique name which is followed by the IUPAC (International Union of Pure and Applied Chemistry). For alkane also, there is a particular rule for IUPAC nomenclature. The rules are −
- The name has to end with suffix "−ane" for all alkanes.
- The longest continuous carbon chain that contains the functional group will be treated as the main chain.
- Number the carbons in the longest carbon chain. As per as the no of carbon present in the longest chain, "Pent (for 5)", "Hept (for 6)" this word will be added before "ane".
- The branched groups present in the longest carbon chain beside the main chain should be named by the number of carbon atoms present in the branched group. These groups will end with "-yl" (e.g.- ethyl, methyl) at their end.
- The position of the group on the main carbon chain and it should be mentioned (e.g. 2,3 -).
- Numbering of the main chain should start from the side having smallest distance with most priority brunch (priority depends on alphabetical order of the brunch).
- The brunched groups should be listed before the name of the main carbon chain in alphabetical order (ignoring prefix like di/tri).
- Combine the elements of the name into a single word in the following order:
- Branched groups in alphabetical order (ignoring prefixes).
- Prefix of main chain
- End with "-ane" suffix.
Answer to Problem 45P
3,3-diethylhexane
Explanation of Solution
The longest carbon chain has 8 carbon.
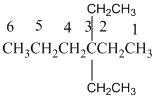
And there are two ethyl groups in the no. 3 carbon. As the branch groups have CH2CH3, it is called 'ethyl'.
So, following by the above mention rule, the name of the alkane is 3,3 (the position of the branch) + diethyl (the name of the branch, 'di' as two ethyl are present)+ hex( 6 carbon in main chain)+ "-ane"= 3,3-diethylhexane.
(d)
Interpretation:
The IUPAC nomenclature of Alkanes having skeletal structure needs to be determined.
Concept Introduction:
Every organic compound has its own unique name which is followed by the IUPAC (International Union of Pure and Applied Chemistry). For alkane also, there is a particular rule for IUPAC nomenclature. The rules are −
- The name has to end with suffix "−ane" for all alkanes.
- The longest continuous carbon chain that contains the functional group will be treated as the main chain.
- Number the carbons in the longest carbon chain. As per as the no of carbon present in the longest chain, "Pent (for 5)", "Hept (for 6)" this word will be added before "ane".
- The branched groups present in the longest carbon chain beside the main chain should be named by the number of carbon atoms present in the branched group. These groups will end with "-yl" (e.g.- ethyl, methyl) at their end.
- The position of the group on the main carbon chain and it should be mentioned (e.g. 2,3 -).
- Numbering of the main chain should start from the side having smallest distance with most priority brunch (priority depends on alphabetical order of the brunch).
- The brunched groups should be listed before the name of the main carbon chain in alphabetical order (ignoring prefix like di/tri).
- Combine the elements of the name into a single word in the following order:
- Branched groups in alphabetical order (ignoring prefixes).
- Prefix of main chain
- End with "-ane" suffix.
Answer to Problem 45P
4-ethyl-2,6-dimethyloctane
Explanation of Solution
The longest carbon chain has 8 carbon.
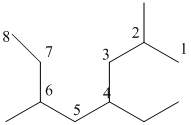
And there are two methyl groups in the no. 2 and 6 carbon, one ethyl group in no. 4carbon. As the branch groups have CH3and CH2CH3, they are called 'methyl' and 'ethyl' simultaneously.
So, following by the above mention rule, the name of the alkane is 4 (the position of the branch having alphabetically order 1st) +ethyl (the name of the branch) + 2,6 (position of 2nd brunch) + dimethyl( name of the brunch and 'di' as two methyl are present)+ oct ( 8 carbon in main chain)+ "-ane"= 4-ethyl-2,6-dimethyloctane
(e)
Interpretation:
The IUPAC nomenclature of alkanes having different brunches in different position needs to be determined.
Concept Introduction:
Every organic compound has its own unique name which is followed by the IUPAC (International Union of Pure and Applied Chemistry). For alkane also, there is a particular rule for IUPAC nomenclature. The rules are −
- The name has to end with suffix "−ane" for all alkanes.
- The longest continuous carbon chain that contains the functional group will be treated as the main chain.
- Number the carbons in the longest carbon chain. As per as the no of carbon present in the longest chain, "Pent (for 5)", "Hept (for 6)" this word will be added before "ane".
- The branched groups present in the longest carbon chain beside the main chain should be named by the number of carbon atoms present in the branched group. These groups will end with "-yl" (e.g.- ethyl, methyl) at their end.
- The position of the group on the main carbon chain and it should be mentioned (e.g. 2,3 -).
- Numbering of the main chain should start from the side having smallest distance with most priority brunch (priority depends on alphabetical order of the brunch).
- The brunched groups should be listed before the name of the main carbon chain in alphabetical order (ignoring prefix like di/tri).
- Combine the elements of the name into a single word in the following order:
- Branched groups in alphabetical order (ignoring prefixes).
- Prefix of main chain
- End with "-ane" suffix.
Answer to Problem 45P
6-ethyl-2-methyloctane
Explanation of Solution
The longest carbon chain has 8 carbon.
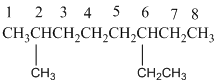
And there is one methyl group in the no. 2 carbon, one ethyl group in no. 6 carbon. As the branch groups have CH3and CH2CH3, they are called 'methyl' and 'ethyl' simultaneously.
So, following by the above mention rule, the name of the alkane is 6 (the position of the branch having alphabetically order 1st) + ethyl (the name of the branch) + 2 (position of 2nd brunch) + methyl( name of the brunch )+ oct ( 8 carbon in main chain)+ "-ane"= 6-ethyl-2-methyloctane
(f)
Interpretation:
The IUPAC nomenclature of Alkanes having lots of groups present in different position needs to be determined.
Concept Introduction:
Every organic compound has its own unique name which is followed by the IUPAC (International Union of Pure and Applied Chemistry). For alkane also, there is a particular rule for IUPAC nomenclature. The rules are −
- The name has to end with suffix "−ane" for all alkanes.
- The longest continuous carbon chain that contains the functional group will be treated as the main chain.
- Number the carbons in the longest carbon chain. As per as the no of carbon present in the longest chain, "Pent (for 5)", "Hept (for 6)" this word will be added before "ane".
- The branched groups present in the longest carbon chain beside the main chain should be named by the number of carbon atoms present in the branched group. These groups will end with "-yl" (e.g.- ethyl, methyl) at their end.
- The position of the group on the main carbon chain and it should be mentioned (e.g. 2,3 -).
- Numbering of the main chain should start from the side having smallest distance with most priority brunch (priority depends on alphabetical order of the brunch).
- The brunched groups should be listed before the name of the main carbon chain in alphabetical order (ignoring prefix like di/tri).
- Combine the elements of the name into a single word in the following order:
- Branched groups in alphabetical order (ignoring prefixes).
- Prefix of main chain
- End with "-ane" suffix.
Answer to Problem 45P
3,3,9,9-tetramethylundecane
Explanation of Solution
The longest carbon chain has 8 carbon.
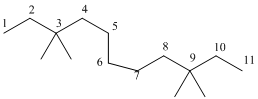
And there are four methyl groups in the no. 3 and 9 carbon. As the branch groups have CH3, it is called 'methyl'.
So, following by the above mention rule, the name of the alkane is 3,3,9,9 (the position of the branch) + tetramethyl ( name of the brunch and 'tetra' as four methyl are present)+ undacane ( 11 carbon in main chain)+ "-ane"= 3,3,9,9-tetramethylundecane.
Want to see more full solutions like this?
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Choose the correct structure for each of the following compounds. a. an alcohol with molecular formula C₂H6O O CH3 CH₂-O-CH3 O CH3-C-H OCH3-CH₂-OH O CH3-O-CH3 b. an aldehyde with molecular formula C3H$O ů OCH, CCH, O CH3-CH₂-O-CH3 OCH3 CH₂-CH OH CH3-CH-CH3arrow_forwardQUESTION 2 The following is a proposed synthetic strategy that uses one starting alcohol in two separate single-step reactions (I and II) to make a pair of diene and dienophile which together undergo a Diels-Alder reaction to give a Diels-Alder product. The Diels-Alder product is subsequently converted to the final ether in a three-step synthesis (III, IV and V). Identify the reagent(s) from Table I required for each of those three sysnthese and place the number (A1, A2 reagent(s) in the given box right next to the corresponding step numbers (I to V) of the synthesis. DO NOT fill in any box with more than one number. (Note: This question is different from the previous synthesis question which allows two numbers for each box). You will be graded based on the number in each box. Do not fill in any single box with more than one number. Do not leave any box blank. B1, B2..., or C1, C2 ...) that represents the (I) diene OH (III, IV and V) Diesl-Alder product (II) dienophile Table I Number…arrow_forwardA. Classification of Hydrocarbons. H H 6. H-C=C-C-C-H H H 2. H-C-Ċ-H H HHH H-C=C-C=C-H 8. CH CH; CH CH-CH,-CH, CH: CH, CH, CH, 10.arrow_forward
- 1. Given below are the structures of five hydrocarbons: Culo (244 H H H H H CH3 H-C-C- HHHH H-C-C-C-H H-C-C-C-C-H C=C H H C-H H. H H A C Explain why all five compounds above are hydrocarbons. because they have eleuets carbon hudrogen only. c 2. Classify the compounds A to D as either alkanes, alkenes, alkyne or aromatic. A. a Kunes B. elkanes C.alkenes D. alkanes С. HICIH Oarrow_forwardThis compound has the common name acetone. What is its proper systematic name? O C-C- C-C сarrow_forwardCH HCI, H₂O Hg(OAc)2 H₂O H₂SO4 H₂O NC. CN LIAIH4, ether KCN H₂Oarrow_forward
- Calculate the enthalpy for the following reaction. CH₂=CH₂ H-CI DH CH2=CH2, 62 kcal/mole H-CI, 103 kcal/mole CH3CH2-H, 101 kcal/mole CH3CH2-CI, 85 kcal/mole CH₂ CH₂ f;arrow_forwardW What is the correct IUPAC name for the compound shown here? C # 3 e Oll 4 F4 2,2- 2- LL % de in r O 5 Question 11 of 49 t tetra ISO sec- di Σ F6 3- 4- 2,3- 3,4- < 60 tri GO DELL A y F7 & 7 4 u F8 * CO 8 F9 ( 9 F10 0 0 F11 Р 8 F12 Mar 24 E [ 8 deletearrow_forwardWrite the common (not systematic) name of each organic molecule. structure CH3 CH3 CH3—CH—NH–CH–CH3 CH₂ - CH3 CH3-CH₂-CH2-N-CH₂ - CH3 CH3 CH3 CH3-CH-N-CH3 name 7 Пarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





