
Concept explainers
(a)
Interpretation:
The product of the given reaction should be given.
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Bromination:

2-methyl propane undergoes radical bromination which yields the 2-bromo-2-methylpropane.because bromination will occur where the tertiary radical is present.(Bromination reactions are more selective reaction).
The mechanism of monobromination of ethane (as an example) includes three steps,
- (i) Initiation
- (ii) Propagation
- (iii) Termination
Thus the mechanism of monobromination of ethane is shown below,
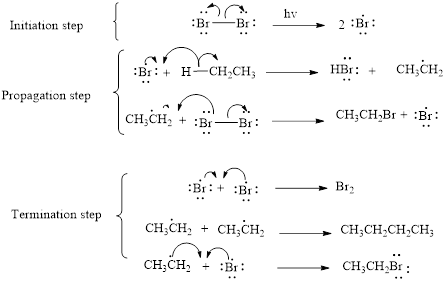
In a halogenation reaction, one or more halogen atoms are introduced into an organic compound. Generally, these reactions are initiated in the presence of light or heat.
Bromination will occur on tertiary radical than the secondary than primary radical, tertiary radical is more stable radical than the other radicals.
(a)
Answer to Problem 12P
3-methyl hexane undergoes radical bromination and yields the 3-bromo-3-methylhexane which is shown below
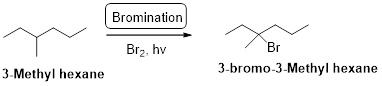
Explanation of Solution
3-methyl hexane undergoes radical bromination and yields the 3-bromo-3-methylhexane according to the above mentioned mechanism steps. The reaction is shown below,
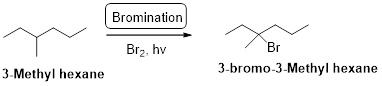
(b)
Interpretation:
The product of the given reaction should be given.
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Chlorination:
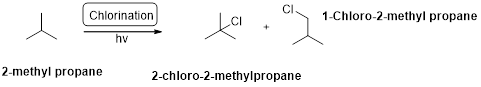
2-methyl propane undergoes radical chlorination and yields the 2-bromo-2-methylpropane and 1-bromo-2-methyl propane.
The mechanism of monochlorination of ethane includes three steps,
- (i) Initiation
- (ii) Propagation
- (iii) Termination
The mechanism of monochlorination of ethane is shown below,
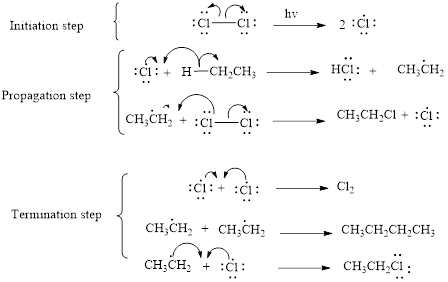
In a halogenation reaction, one or more halogen atoms are introduced into an organic compound. Generally, these reactions are initiated in the presence of light or heat.
(b)
Explanation of Solution
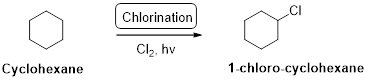
Cyclohexane undergoes radical chlorination; all the carbons in cyclohexane are secondary. Therefore, it yields the 1-chloro cyclohexane according to the above mentioned mechanism steps. And the reaction is shown above.
(c)
Interpretation:
The product of the given reaction should be given.
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Chlorination:
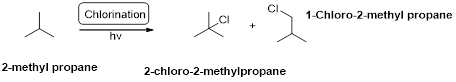
2-methyl propane undergoes radical chlorination and yields the 2-bromo-2-methylpropane and 1-bromo-2-methyl propane.
The mechanism of monochlorination of ethane includes three steps,
- (i) Initiation
- (ii) Propagation
- (iii) Termination
The mechanism of monochlorination of ethane is shown below,

In a halogenation reaction, one or more halogen atoms are introduced into an organic compound. Generally, these reactions are initiated in the presence of light or heat.
(c)
Explanation of Solution
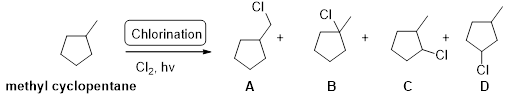
Methyl cyclopentane undergoes radical chlorination; the carbons in cyclopentane are secondary and primary. Therefore, it yields the four types of chlorocyclopentane according to the above mentioned mechanism steps. And the reaction is shown below
Want to see more full solutions like this?
Chapter 14 Solutions
Essential Organic Chemistry (3rd Edition)
- Identify the missing reagents a-f in the following scheme:arrow_forwardDraw the major organic product when this compound is treated with NBS and heat: H3C -CH3arrow_forward7.61 Draw a stepwise mechanism for the following reaction that illustrates how two substitution products are formed. Explain why 1-bromohex-2- ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reaction conditions, even though it is a 1 ° alkyl halide. OCH3 CH;OH Br OCH3 + HBr 1-bromohex-2-enearrow_forward
- Draw a stepwise mechanism for the following reaction. Part I out of 4 Nucleophilic substitution of H for C1:arrow_forwardDraw a structure for the product of nucleophilic substitution obtained on solvolysis of tert-butyl bromide in methanol, and arrange the correct mechanism for its formation. Be sure to answer all parts.arrow_forwardHydroxy aldehydes A and B readily cyclize to form hemiacetals. Draw the stereoisomers formed in this reaction from both A and B. Explain why this process gives an optically inactive product mixture from A, and an optically active product mixture from B. CHO CHO но A Barrow_forward
- Draw the starting structure that would lead to the major product shown under the provided conditions. Drawing 1. NaNH2 2. CH3CH2CH2Br > aarrow_forwardRank the following alkenes in order of increasing stability: A вarrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. CH3COCl, AlCl3arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

