
Interpretation:
The solubility of naphthalene (1) in carbon dioxide (2) at a given temperature and pressure should be estimated and compare the results with given graph and comment on them and differences should be discussed at P1sat =0.0102 bar at 80oC.
Concept Introduction:
The solubility of solid in the solvent carbon dioxide is calculated by following formula which is equation (15.28)
And for naphthalene at infinite dilution in CO2,
Answer to Problem 15.19P
The solubility of naphthalene increases then after some time remains constant. Solubility is affected by the temperature.
Explanation of Solution
Given information:
It is given that the operating conditions are
SVE is given with
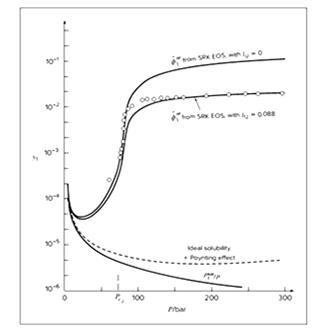
Solubility graph of naphthalene (1) in carbon dioxide (2) is given as
For simplicity, considering for naphthalene at infinite dilution in carbon dioxide, the fugacity coefficient in equation (1) is at infinite dilution, hence from equation (1) function F1 is
Since it is given that vapor pressure is very small and the saturated vapor is for practical purposes an ideal gas, hence at this condition
Hence equation (1) becomes
Hence solubility is
Where, 1 is used for naphthalene and 2 will use for carbon dioxide.
From equation (2)
Where
For vapors
And it is given that
For the calculation of
From SRK equation for the calculation of parameters assigned to equation of state for vapors is
And the characteristics properties of pure naphthalene and carbon dioxide is given in Appendix B, Table B.1
For naphthalene
For pure carbon dioxide
One by one solving each quantity
For carbon dioxide
And
Therefore,
And
For naphthalene
And,
Put the values in equation (2)
Therefore, solubilities at different pressure
Put the values of pressure and hence solubilities are
| P1 | β2 | Z2 | I2 | ln(f1) | f1 | y1 |
| 20 | 0.0202 | 0.944 | 0.021173 | -0.47849 | 0.619718 | 0.000895792 |
| 40 | 0.0404 | 0.887 | 0.04454 | -0.98525 | 0.373347 | 0.000809263 |
| 60 | 0.0606 | 0.828 | 0.070634 | -1.52951 | 0.216642 | 0.001012043 |
| 80 | 0.0808 | 0.768 | 0.100034 | -2.11397 | 0.120758 | 0.001482243 |
| 100 | 0.101 | 0.709 | 0.133179 | -2.73612 | 0.064822 | 0.002404564 |
| 120 | 0.1212 | 0.653 | 0.170253 | -3.39027 | 0.0337 | 0.004195473 |
| 140 | 0.1414 | 0.605 | 0.210033 | -4.0444 | 0.01752 | 0.007529222 |
| 160 | 0.1616 | 0.569 | 0.249986 | -4.65524 | 0.009512 | 0.013209077 |
| 180 | 0.1818 | 0.546 | 0.287407 | -5.18826 | 0.005582 | 0.021779138 |
| 200 | 0.202 | 0.535 | 0.320321 | -5.62472 | 0.003608 | 0.033011622 |
| 220 | 0.2222 | 0.533 | 0.348461 | -5.97226 | 0.002548 | 0.046242035 |
| 240 | 0.2424 | 0.536 | 0.373106 | -6.25881 | 0.001914 | 0.061451133 |
| 260 | 0.2626 | 0.542 | 0.395079 | -6.50057 | 0.001503 | 0.07863101 |
| 280 | 0.2828 | 0.551 | 0.414259 | -6.69709 | 0.001235 | 0.096735253 |
| 300 | 0.303 | 0.561 | 0.431852 | -6.86828 | 0.00104 | 0.116627234 |
The graph between pressure and the solubilities is
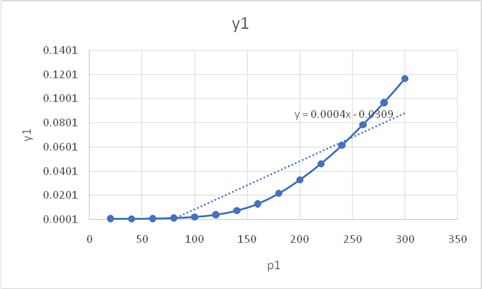
On comparison of graph from the given graph
From the found graph, one can clearly conclude that the solubility of the naphthalene is constant initially at low pressure but as pressure increases its solubility also increases reaches up to 0.12 but after very high pressure above 300 bar it remains constant. The given graph of solubility v/s pressure shows that at
The temperature of graph found is
The solubility of naphthalene increases then after some time remains constant. Solubility is affected by the temperature.
Want to see more full solutions like this?
Chapter 15 Solutions
Introduction to Chemical Engineering Thermodynamics
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





