
Concept explainers
(a)
Interpretation: The most stable radical that can result from cleavage of
Concept introduction: A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.2P
The most stable radical that can result from cleavage of
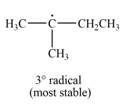
Explanation of Solution
The given species is,
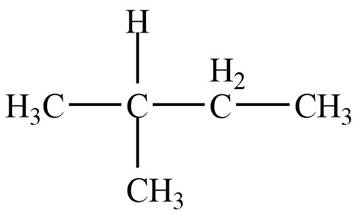
Figure 1
Three types of radicals can be formed by the cleavage of
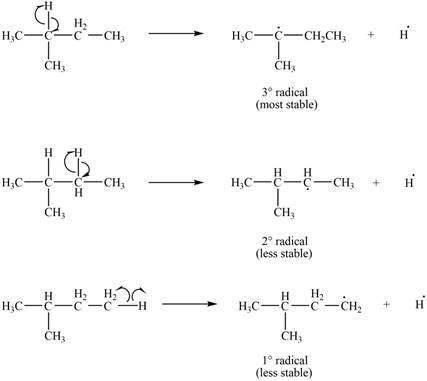
Figure 2
The number of alkyl substitutents increases, the stability of radical increases. The order of stability is
The most stable radical that can result from cleavage of
(b)
Interpretation: The most stable radical that can result from cleavage of
Concept introduction: A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.2P
The most stable radical that can result from cleavage of
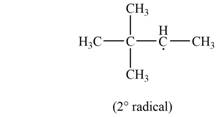
Explanation of Solution
The given species is,
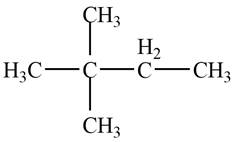
Figure 3
Two types of radicals can be formed from cleavage of
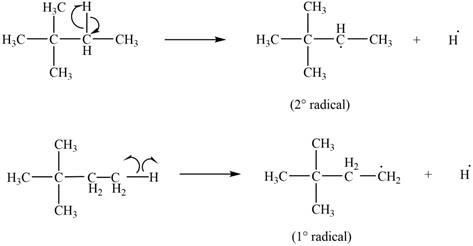
Figure 4
The number of alkyl substitutents increases, the stability of radical increases. The order of stability is
The most stable radical that can result from cleavage of
(c)
Interpretation: The most stable radical that can result from cleavage of
Concept introduction: A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.2P
The most stable radical that can results from the cleavage of
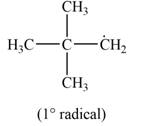
Explanation of Solution
The given species is,
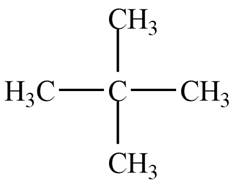
Figure 5
Only
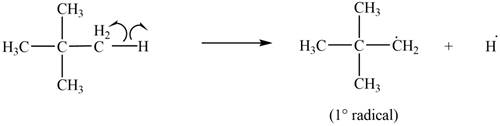
Figure 6
The most stable radical that can result from cleavage of
(d)
Interpretation: The most stable radical that can result from cleavage of
Concept introduction: A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.2P
The most stable radical that can result from cleavage of
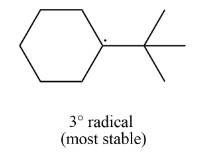
Explanation of Solution
The given species is,
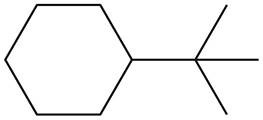
Figure 7
Two types of radicals can be formed from cleavage of
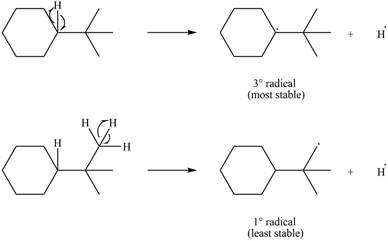
Figure 8
The number of alkyl substitutents increases, the stability of radical increases. The order of stability is
The most stable radical that can result from cleavage of
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry
- Choose the correct product for each reaction. A. B. C. D. PPh3 H PPh3 || I ။arrow_forwardExamine the reactant and product for each reaction. Label each transformation as an addition, elimination, or substitution. Br А. Br ОН elimination addition С. substitution D. B.arrow_forwardComplete the fourth resonance structure to determine which bond is shortest? Which compound will react fourth fastest with B-eBr ? d. e. F 14. Complete the fourth resonance structure to determine which bond is shortest? 15. Which compound will react fourth fastest with Br₂/FeBr3? a. b. d. -OH b darrow_forward
- 1. Which among these can make a molecule nucleophilic? a.double bondsb.positive chargec. incomplete octet 2. Which among these can make a molecule electrophilic? a.Triple bondsb.positive chargec. radicalsarrow_forwardWhich B carbon in the molecule below would be used to form the Zaitsev product? Br a. A b.B C. C d. D e. E O f. F ajarrow_forward5. Consider the reaction below and answer the following questions + OH + H₂O a. Draw in ALL missing lone pair electrons as well as electron movement arrows on the reactant side Identify the nucleophile and electrophile on the reactant side b. c. Identify which side of the equilibrium will dominate. d. Draw resonance structures for the organic compound on the product side CHEM 241-Group05. Ormord d ****arrow_forward
- Consider this step in a radical reaction: Br Br: ?arrow_forwardWhich of the following is not stable? a. ?H b. Li c. ®B d. 14N Aarrow_forwardH₂C CH₂ 2 c CH₂ 1. Reaction A is a radical process. The part of the mechanism that is represented above is called [blank1] would immediately follow [blank2] A. a. nucleophilic attack; hydrogen abstraction b. homolytic attack; hydrogen abstraction c. hydrogen abstraction; homolytic cleavage d. nucleophilic attack; homolytic cleavage 2. Radical mechanisms begins by which of the following events: a) homolytic cleavage b) heterolytic cleavage c) extreme heating or exposure to light irradiation. d) nucleophilic attack 3. Rank the following radicals from most stable to least stable: CH H₂C A CH₂ a) D > C>A>B b) B>D>A>C c) D> B>A>C d) C>A>B>D H₂C B CH₂ H₂C" CH₂ C CH3 H₂C CH D CH₂arrow_forward
- a. 5. Draw the product(s) of each addition reaction b. d. دھ HBr H2SO4, H₂O Bra, H2O a. 9-BBN b. H2O2, NaOHarrow_forwardComplete the sentence. Hyperconjugation describes how a carbocation is stabilized when the electrons of ____ donate electron density into____. A. a pi bond / an eclipsed C:H bond B. radical / the adjacent pi bond C. a double bond / the positive carbon D. a sigma bond / an empty p orbitalarrow_forward1. Products A and B combine to make up over 90% of the product mixture. H-CI CI В D ČI A a. For each of the structure A-D, attach an H atom to the carbon that in fact added an H b. Classify each of the four structures as either a 1,2 or 1,4 addition product c. Draw the resonance structures for the cation that leads to both product A+B, and also draw the resonance structure for the cation that leads to both product C+D. d. Draw the mechanism for the formation of products A and B above.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





